Melanin

Melanin ![]() i/ˈmɛlənɪn/ (Greek: μέλας - melas, "black, dark") is a broad term for a group of natural pigments found in most organisms (arachnids are one of the few groups in which it has not been detected). Melanin is a derivative of the amino acid tyrosine, however it is not itself made of amino acids and is not a protein. The pigment is produced in a specialized group of cells known as melanocytes.
i/ˈmɛlənɪn/ (Greek: μέλας - melas, "black, dark") is a broad term for a group of natural pigments found in most organisms (arachnids are one of the few groups in which it has not been detected). Melanin is a derivative of the amino acid tyrosine, however it is not itself made of amino acids and is not a protein. The pigment is produced in a specialized group of cells known as melanocytes.
There are three basic types of melanin: eumelanin, pheomelanin, and neuromelanin. The most common type is eumelanin, and is produced in 'black' and 'brown' subtypes. Pheomelanin is a cysteine-containing red-brown polymer of benzothiazine units largely responsible for red hair and freckles. Neuromelanin is found in the brain, though its function remains obscure.
The production of melanin is called melanogenesis. In the skin, melanogenesis occurs after exposure to UV radiation, causing the skin to visibly tan. Melanin is an effective absorber of light; the pigment is able to dissipate over 99.9% of absorbed UV radiation.[1] Because of this property, melanin is thought to protect skin cells from UVB radiation damage, reducing the risk of cancer. Furthermore, though exposure to UV radiation is associated with increased risk of malignant melanoma, a cancer of the melanocytes, studies have shown a lower incidence for skin cancer in individuals with more concentrated melanin, i.e. darker skin tone. Nonetheless, the relationship between skin pigmentation and photoprotection is still being clarified.[2]
Humans

In humans, melanin is the primary determinant of skin color. It is also found in hair, the pigmented tissue underlying the iris of the eye, and the stria vascularis of the inner ear. In the brain, tissues with melanin include the medulla and pigment-bearing neurons within areas of the brainstem, such as the locus coeruleus and the substantia nigra. It also occurs in the zona reticularis of the adrenal gland.
The melanin in the skin is produced by melanocytes, which are found in the basal layer of the epidermis. Although, in general, human beings possess a similar concentration of melanocytes in their skin, the melanocytes in some individuals and ethnic groups more frequently or less frequently express the melanin-producing genes, thereby conferring a greater or lesser concentration of skin melanin. Some individual animals and humans have very little or no melanin synthesis in their bodies, a condition known as albinism.
Because melanin is an aggregate of smaller component molecules, there are many different types of melanin with differing proportions and bonding patterns of these component molecules. Both pheomelanin and eumelanin are found in human skin and hair, but eumelanin is the most abundant melanin in humans, as well as the form most likely to be deficient in albinism.
Eumelanin
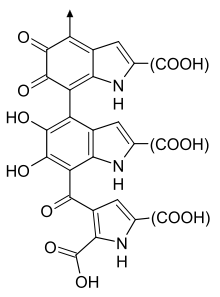
Eumelanin polymers have long been thought to comprise numerous cross-linked 5,6-dihydroxyindole (DHI) and 5,6-dihydroxyindole-2-carboxylic acid (DHICA) polymers. The two types are recognized: black and brown. Black melanin is darker than brown. A small amount of black eumelanin in the absence of other pigments causes grey hair. A small amount of brown eumelanin in the absence of other pigments causes yellow (blond) color hair.
Pheomelanin
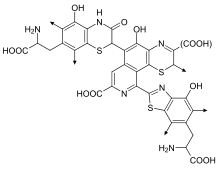
Pheomelanin imparts a pink to red hue and, thus, is found in particularly large quantities in red hair.[3] Pheomelanin is particularly concentrated in the lips, nipples, glans of the penis, and vagina.[4] In chemical terms, pheomelanin differs from eumelanin in that its oligomer structure incorporates benzothiazine and benzothiazole units that are produced,[5] instead of DHI and DHICA, when the amino acid L-cysteine is present.
Neuromelanin
Neuromelanin (NM) is a dark polymer pigment produced in specific populations of catecholaminergic neurons in the brain. Humans have the most number of NM, while they are in lesser amount in other non-human primates, and totally absent in lower species.[6] However, the biological function remains unknown, although human NM has been shown to efficiently bind transition metals such as iron, as well as other potentially toxic molecules. Therefore, it may play crucial roles in apoptosis and the related Parkinson's disease.[7]
Other organisms
Melanins have very diverse roles and functions in various organisms. A form of melanin makes up the ink used by many cephalopods (see cephalopod ink) as a defense mechanism against predators. Melanins also protect microorganisms, such as bacteria and fungi, against stresses that involve cell damage such as UV radiation from the sun and reactive oxygen species. Melanin also protects against damage from high temperatures, chemical stresses (such as heavy metals and oxidizing agents), and biochemical threats (such as host defenses against invading microbes).[8] Therefore, in many pathogenic microbes (for example, in Cryptococcus neoformans, a fungus) melanins appear to play important roles in virulence and pathogenicity by protecting the microbe against immune responses of its host. In invertebrates, a major aspect of the innate immune defense system against invading pathogens involves melanin. Within minutes after infection, the microbe is encapsulated within melanin (melanization), and the generation of free radical byproducts during the formation of this capsule is thought to aid in killing them.[9] Some types of fungi, called radiotrophic fungi, appear to be able to use melanin as a photosynthetic pigment that enables them to capture gamma rays[10] and harness its energy for growth.[11]
The black feathers of birds owe their color to melanin; they are much more readily degraded by bacteria than white feathers, or those containing other pigments such as carotenes.[12]
In some mice, melanin is used slightly differently. For instance, in Agouti mice, the hair appears brown because of alternation between black eumelanin production and a yellow variety of pheomelanin. The hairs are actually banded black and yellow, and the net effect is the brown color of most mice. Some genetic irregularities can produce either fully black or fully yellow mice.
Catechol melanins are plant melanins.
Biosynthetic pathways

.svg.png)


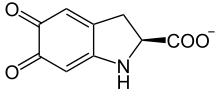
The first step of the biosynthetic pathway for both eumelanins and pheomelanins is catalysed by tyrosinase:
Dopaquinone can combine with cysteine by two pathways to benzothiazines and pheomelanins
- Dopaquinone + cysteine → 5-S-cysteinyldopa → benzothiazine intermediate → pheomelanin
- Dopaquinone + cysteine → 2-S-cysteinyldopa → benzothiazine intermediate → pheomelanin
Also, dopaquinone can be converted to leucodopachrome and follow two more pathways to the eumelanins
- Dopaquinone → leucodopachrome → dopachrome → 5,6-dihydroxyindole-2-carboxylic acid → quinone → eumelanin
- Dopaquinone → leucodopachrome → dopachrome → 5,6-dihydroxyindole → quinone → eumelanin
Detailed metabolic pathways can be found in the KEGG database (see External links).
Microscopic appearance
Melanin is brown, non-refractile, and finely granular with individual granules having a diameter of less than 800 nanometers. This differentiates melanin from common blood breakdown pigments, which are larger, chunky, and refractile, and range in color from green to yellow or red-brown. In heavily pigmented lesions, dense aggregates of melanin can obscure histologic detail. A dilute solution of potassium permanganate is an effective melanin bleach.
Genetic disorders and disease states
Melanin deficiency has been connected for some time with various genetic abnormalities and disease states.
There are approximately nine different types of oculocutaneous albinism, which is mostly an autosomal recessive disorder. Certain ethnicities have higher incidences of different forms. For example, the most common type, called oculocutaneous albinism type 2 (OCA2), is especially frequent among people of black African descent. It is an autosomal recessive disorder characterized by a congenital reduction or absence of melanin pigment in the skin, hair, and eyes. The estimated frequency of OCA2 among African-Americans is 1 in 10,000, which contrasts with a frequency of 1 in 36,000 in white Americans.[13] In some African nations, the frequency of the disorder is even higher, ranging from 1 in 2,000 to 1 in 5,000.[14] Another form of Albinism, the "yellow oculocutaneous albinism", appears to be more prevalent among the Amish, who are of primarily Swiss and German ancestry. People with this IB variant of the disorder commonly have white hair and skin at birth, but rapidly develop normal skin pigmentation in infancy.[14]
Ocular albinism affects not only eye pigmentation but visual acuity, as well. People with albinism typically test poorly, within the 20/60 to 20/400 range. In addition, two forms of albinism, with approximately 1 in 2700 most prevalent among people of Puerto Rican origin, are associated with mortality beyond melanoma-related deaths.
Mortality also is increased in patients with Hermansky-Pudlak syndrome and Chediak-Higashi syndrome. Patients with Hermansky-Pudlak syndrome have a bleeding diathesis secondary to platelet dysfunction and also experience restrictive lung disease (pulmonary fibrosis), inflammatory bowel disease, cardiomyopathy, and renal disease. Patients with Chediak-Higashi syndrome are susceptible to infection and also can develop lymphofollicular malignancy.—[14]
The role that melanin deficiency plays in such disorders remains under study.
The connection between albinism and deafness is well known, though poorly understood. In his 1859 treatise On the Origin of Species, Charles Darwin observed that "cats which are entirely white and have blue eyes are generally deaf".[15] In humans, hypopigmentation and deafness occur together in the rare Waardenburg's syndrome, predominantly observed among the Hopi in North America.[16] The incidence of albinism in Hopi Indians has been estimated as approximately 1 in 200 individuals. It is interesting to note that similar patterns of albinism and deafness have been found in other mammals, including dogs and rodents. However, a lack of melanin per se does not appear to be directly responsible for deafness associated with hypopigmentation, as most individuals lacking the enzymes required to synthesize melanin have normal auditory function.[17] Instead the absence of melanocytes in the stria vascularis of the inner ear results in cochlear impairment,[18] though why this is, is not fully understood.
In Parkinson's disease, a disorder that affects neuromotor functioning, there is decreased neuromelanin in the substantia nigra and locus coeruleus as consequence of specific dropping out of dopaminergic and noradrenergic pigmented neurons. This results in diminished dopamine and norepinephrine synthesis. While no correlation between race and the level of neuromelanin in the substantia nigra has been reported, the significantly lower incidence of Parkinson's in blacks than in whites has "prompt[ed] some to suggest that cutaneous melanin might somehow serve to protect the neuromelanin in substantia nigra from external toxins.".[19] Also see Nicolaus[20] review article on the function of neuromelanins.
In addition to melanin deficiency, the molecular weight of the melanin polymer may be decreased by various factors such as oxidative stress, exposure to light, perturbation in its association with melanosomal matrix proteins, changes in pH, or in local concentrations of metal ions. A decreased molecular weight or a decrease in the degree of polymerization of ocular melanin has been proposed to turn the normally anti-oxidant polymer into a pro-oxidant. In its pro-oxidant state, melanin has been suggested to be involved in the causation and progression of macular degeneration and melanoma.[21] Rasagiline, an important monotherapy drug in Parkinson's disease, has melanin binding properties, and melanoma tumor reducing properties.[22]
Higher eumelanin levels also can be a disadvantage, however, beyond a higher disposition toward vitamin D deficiency. Dark skin is a complicating factor in the laser removal of port-wine stains. Effective in treating white skin, in general, lasers are less successful in removing port-wine stains in people of Asian or African descent. Higher concentrations of melanin in darker-skinned individuals simply diffuse and absorb the laser radiation, inhibiting light absorption by the targeted tissue. In similar manner, melanin can complicate laser treatment of other dermatological conditions in people with darker skin.
Freckles and moles are formed where there is a localized concentration of melanin in the skin. They are highly associated with pale skin.
Nicotine has an affinity for melanin-containing tissues because of its precursor function in melanin synthesis or its irreversible binding of melanin. This has been suggested to underlie the increased nicotine dependence and lower smoking cessation rates in darker pigmented individuals.[23]
Human adaptation
Melanocytes insert granules of melanin into specialized cellular vesicles called melanosomes. These are then transferred into the other skin cells of the human epidermis. The melanosomes in each recipient cell accumulate atop the cell nucleus, where they protect the nuclear DNA from mutations caused by the ionizing radiation of the sun's ultraviolet rays. In general, people whose ancestors lived for long periods in the regions of the globe near the equator have larger quantities of eumelanin in their skins. This makes their skins brown or black and protects them against high levels of exposure to the sun, which more frequently results in melanomas in lighter-skinned people.
With humans, exposure to sunlight stimulates the skin to produce vitamin D. Because high levels of cutaneous melanin act as a natural sun screen, dark skin can be a risk factor for vitamin D deficiency in regions of the Earth known as cool temperate zones, i.e., above 36 degrees latitude in the Northern hemisphere and below 36 degrees in the Southern hemisphere. As a result of this, health authorities in Canada and the USA have issued recommendations for people with darker complexions (including people of southern European descent) to consume between 1000-2000 IU (International Units) of vitamin D, daily, autumn through spring.
The most recent scientific evidence indicates that all humans evolved in Africa,[24] then populated the rest of the world through successive radiations. It seems likely that the first modern humans had relatively large numbers of eumelanin-producing melanocytes. In accordance, they had darker skin as with the indigenous people of Africa today. As some of these original peoples migrated and settled in areas of Asia and Europe, the selective pressure for eumelanin production decreased in climates where radiation from the sun was less intense. Of the two common gene variants known to be associated with pale human skin, Mc1r[25] does not appear to have undergone positive selection, while SLC24A5[26] has.
As with peoples having migrated northward, those with light skin migrating toward the equator acclimatize to the much stronger solar radiation. Most people's skin darkens when exposed to UV light, giving them more protection when it is needed. This is the physiological purpose of sun tanning. Dark-skinned people, who produce more skin-protecting eumelanin, have a greater protection against sunburn and the development of melanoma, a potentially deadly form of skin cancer, as well as other health problems related to exposure to strong solar radiation, including the photodegradation of certain vitamins such as riboflavins, carotenoids, tocopherol, and folate.
Melanin in the eyes, in the iris and choroid, helps protect them from ultraviolet and high-frequency visible light; people with gray, blue, and green eyes are more at risk for sun-related eye problems. Further, the ocular lens yellows with age, providing added protection. However, the lens also becomes more rigid with age, losing most of its accommodation — the ability to change shape to focus from far to near — a detriment due probably to protein crosslinking caused by UV exposure.
Recent research by J.D. Simon et al.[27] suggests that melanin may serve a protective role other than photoprotection. Melanin is able to effectively ligate metal ions through its carboxylate and phenolic hydroxyl groups, in many cases much more efficiently than the powerful chelating ligand ethylenediaminetetraacetate (EDTA). Thus, it may serve to sequester potentially toxic metal ions, protecting the rest of the cell. This hypothesis is supported by the fact that the loss of neuromelanin observed in Parkinson's disease is accompanied by an increase in iron levels in the brain.
Physical properties and technological applications
Evidence exists in support of a highly cross-linked heteropolymer bound covalently to matrix scaffolding melanoproteins.[28] It has been proposed that the ability of melanin to act as an antioxidant is directly proportional to its degree of polymerization or molecular weight.[29] Suboptimal conditions for the effective polymerization of melanin monomers may lead to formation of lower-molecular-weight, pro-oxidant melanin that has been implicated in the causation and progression of macular degeneration and melanoma.[30] Signaling pathways that upregulate melanization in the retinal pigment epithelium (RPE) also may be implicated in the downregulation of rod outer segment phagocytosis by the RPE. This phenomenon has been attributed in part to foveal sparing in macular degeneration.[31]
See also
- Addison's disease
- Albino
- Carotene
- Griscelli syndrome A syndrome characterised by hypopigmentation.
- Human skin color
- Mc1r
- Melanin theory, a pseudoscientific theory that ascribes supernatural abilities to melanin
- Melanism
- Melanizing agents
- Melanogenesis, the increased production of melanin
- Melanoma
- Melanotropin receptor
- Organic semiconductor
- Parkinson's disease
- Risks and benefits of sun exposure
- SLC24A5
- Vitamin D
References
- ↑ Meredith P, Riesz J (February 2004). "Radiative relaxation quantum yields for synthetic eumelanin". Photochemistry and photobiology 79 (2): 211–6. doi:10.1562/0031-8655(2004)079<0211:RCRQYF>2.0.CO;2. ISSN 0031-8655. PMID 15068035.
- ↑ The Protective Role of Melanin Against UV Damage in Human Skin. Retrieved 5 September 2013.
- ↑ V.Krishnaraj, M.D, Skin Layers
- ↑ http://www.metacyc.org/META/NEW-IMAGE?type=COMPOUND&object=CPD-12380
- ↑ Greco, Giorgia; Lucia Panzella, Luisella Verotta, Marco d’Ischia, Alessandra Napolitano (2011). "Uncovering the Structure of Human Red Hair Pheomelanin:Benzothiazolylthiazinodihydroisoquinolines As Key Building Blocks". Journal of Natural Products 74 (4): 675–82. doi:10.1021/np100740n. PMID 21341762.
- ↑ Fedorow H, Tribl F, Halliday G, Gerlach M, Riederer P, Double KL (2005). "Neuromelanin in human dopamine neurons: comparison with peripheral melanins and relevance to Parkinson's disease". Prog Neurobiol 75 (2): 109–124. doi:10.1016/j.pneurobio.2005.02.001. PMID 15784302.
- ↑ Double KL (2006). "Functional effects of neuromelanin and synthetic melanin in model systems". J Neural Transm 113 (6): 751–756. doi:10.1007/s00702-006-0450-5. PMID 16755379.
- ↑ Hamilton AJ, Gomez BL. (2002). "Melanins in fungal pathogens" (PDF). Journal of Medical Microbiology 51 (3): 189–91. PMID 11871612. Retrieved 6 January 2012.
- ↑ Cerenius L, Söderhäll K (April 2004). "The prophenoloxidase-activating system in invertebrates". Immunological reviews 198: 116–26. doi:10.1111/j.0105-2896.2004.00116.x. PMID 15199959.
- ↑ Science News, Dark Power: Pigment seems to put radiation to good use, Week of May 26, 2007; Vol. 171, No. 21 , p. 325 by Davide Castelvecchi
- ↑ Dadachova E, Bryan RA, Huang X, Moadel T, Schweitzer AD, Aisen P, Nosanchuk JD, Casadevall A. (2007). "Ionizing radiation changes the electronic properties of melanin and enhances the growth of melanized fungi". In Rutherford, Julian. PLoS ONE 2 (5): e457. doi:10.1371/journal.pone.0000457. PMC 1866175. PMID 17520016.
- ↑ Grande, Juan Manuel; Negro, Juan José; María Torres, José (2004). "The evolution of bird plumage colouration; a role for feather-degrading bacteria?". Ardeola 51 (2): 375–383.
- ↑ Oculocutaneous Albinism
- ↑ 14.0 14.1 14.2 "Ocular Manifestations of Albinism"
- ↑ Termination of British Library Net internet service
- ↑ OMIM Result
- ↑ Omim - Tyrosinase; Tyr
- ↑ Cable J, Huszar D, Jaenisch R, Steel KP (February 1994). "Effects of mutations at the W locus (c-kit) on inner ear pigmentation and function in the mouse". Pigment Cell Research 7 (1): 17–32. doi:10.1111/j.1600-0749.1994.tb00015.x. PMID 7521050.
- ↑ Lewy Body Disease
- ↑ Nicolaus BJ (2005). "A critical review of the function of neuromelanin and an attempt to provide a unified theory". Med. Hypotheses 65 (4): 791–6. doi:10.1016/j.mehy.2005.04.011. PMID 15949901.
- ↑ Meyskens FL, Farmer P, Fruehauf JP (June 2001). "Redox regulation in human melanocytes and melanoma". Pigment Cell Research 14 (3): 148–54. doi:10.1034/j.1600-0749.2001.140303.x. PMID 11434561.
- ↑ USA (2013-03-25). "Comparison of oral and transdermal admini... [Cutan Ocul Toxicol. 2012] - PubMed - NCBI". Ncbi.nlm.nih.gov. Retrieved 5 September 2013.
- ↑ King G, Yerger VB, Whembolua GL, Bendel RB, Kittles R, Moolchan ET. Link between facultative melanin and tobacco use among African Americans.(2009). Pharmacol Biochem Behav. 92(4):589-96. doi:10.1016/j.pbb.2009.02.011 PMID 19268687
- ↑ Tishkoff SA, Reed FA et al. (April 2009). "The Genetic Structure and History of Africans and African Americans". Science Xpress 324 (5930): 1035–44. doi:10.1126/science.1172257. PMC 2947357. PMID 19407144.
- ↑ Harding RM, Healy E, Ray AJ et al. (April 2000). "Evidence for variable selective pressures at MC1R". American Journal of Human Genetics 66 (4): 1351–61. doi:10.1086/302863. PMC 1288200. PMID 10733465.
- ↑ Lamason RL, Mohideen MA, Mest JR et al. (December 2005). "SLC24A5, a putative cation exchanger, affects pigmentation in zebrafish and humans". Science (New York, New York.) 310 (5755): 1782–6. doi:10.1126/science.1116238. PMID 16357253.
- ↑ Liu Y, Hong L, Kempf VR, Wakamatsu K, Ito S, Simon JD (June 2004). "Ion-exchange and adsorption of Fe(III) by Sepia melanin". Pigment Cell Research 17 (3): 262–9. doi:10.1111/j.1600-0749.2004.00140.x. PMID 15140071.
- ↑ Donatien PD, Orlow SJ (August 1995). "Interaction of melanosomal proteins with melanin". European journal of biochemistry / FEBS 232 (1): 159–64. doi:10.1111/j.1432-1033.1995.tb20794.x. PMID 7556145.
- ↑ Sarangarajan R, Apte SP (2005). "Melanin aggregation and polymerization: possible implications in age-related macular degeneration". Ophthalmic research 37 (3): 136–41. doi:10.1159/000085533. PMID 15867475.
- ↑ Meyskens FL, Farmer PJ, Anton-Culver H (April 2004). "Etiologic pathogenesis of melanoma: a unifying hypothesis for the missing attributable risk". Clinical cancer research : an official journal of the American Association for Cancer Research 10 (8): 2581–3. PMID 15102657.
- ↑ Sarangarajan R, Apte SP (2005). "Melanization and phagocytosis: implications for age related macular degeneration". Molecular vision 11: 482–90. PMID 16030499.
- "Link 4-Melanin 95-97," taken from R.A.Nicolaus,G.Scherillo La Melanina.Un riesame su struttura,proprietà e sistemi, Atti della Accademia Pontaniana, Vol.XLIV,265-287, Napoli 1995.
- Dr. Mohammed O. Peracha, Dean Elloit, and Enrique Garcia-Valenzuela, "Occular Manifestations of Albinism" (Abstract at emedicine.com, Sept. 13, 2005).
External links
| Look up melanin in Wiktionary, the free dictionary. |
| Look up leuco form in Wiktionary, the free dictionary. |