Glutaminase
| glutaminase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
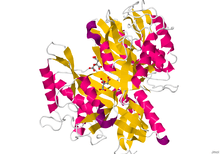 Crystallographic structure of dimeric protein-glutaminase from Chryseobacterium proteolyticum.[1] | |||||||||
| Identifiers | |||||||||
| EC number | 3.5.1.2 | ||||||||
| CAS number | 9001-47-2 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / EGO | ||||||||
| |||||||||
| Glutaminase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 probable glutaminase from bacillus subtilis complexed with 6-diazo-5-oxo-l-norleucine | |||||||||
| Identifiers | |||||||||
| Symbol | Glutaminase | ||||||||
| Pfam | PF04960 | ||||||||
| Pfam clan | CL0013 | ||||||||
| InterPro | IPR015868 | ||||||||
| SCOP | 1mki | ||||||||
| SUPERFAMILY | 1mki | ||||||||
| |||||||||
Glutaminase (EC 3.5.1.2, glutaminase I, L-glutaminase, glutamine aminohydrolase) is an amidohydrolase enzyme that generates glutamate from glutamine. Glutaminase has tissue-specific isoenzymes. Glutaminase has an important role in glial cells.
Glutaminase catalyzes the following reaction:
Glutamine + H2O → Glutamate + NH3
Tissue distribution
Glutaminase is expressed in periportal hepatocytes, where it generates NH3 (ammonia) for urea synthesis, as does glutamate dehydrogenase. Glutaminase is also expressed in the epithelial cells of the renal tubules, where the produced ammonia is excreted as ammonium ions. This excretion of ammonium ions is an important mechanism of renal acid-base regulation. During chronic acidosis, glutaminase is induced in the kidney, which leads to an increase in the amount of ammonium ions excreted. Glutaminase can also be found in the intestines, whereby hepatic portal ammonia can reach as high as 0.26 mM (compared to an arterial blood ammonia of 0.02 mM).
One of the most important roles of glutaminase is found in the axonal terminals of neurons in the central nervous system. Glutamate is the most abundantly used excitatory neurotransmitter in the CNS. After being released into the synapse for neurotransmission, glutamate is rapidly taken up by nearby astrocytes, which convert it to glutamine. This glutamine is then supplied to the presynaptic terminals of the neurons, where glutaminases convert it back to glutamate for loading into synaptic vesicles. Although both "kidney-type" (GLS1) and "liver-type" (GLS2) glutaminases are expressed in brain, GLS2 has been reported to exist only in cellular nuclei in CNS neurons.[2]
Regulation
ADP is the strongest adenine nucleotide activator of glutaminase. Studies have also suggested ADP lowered the K(m) for glutamine and increased the V(max). They found that these effects were increased even more when ATP was present.[3]
Phosphate-activated mitochondrial glutaminase (GLS2) is suggested to be linked with elevated metabolism, decreased intracellular reactive oxygen species (ROS) levels, and overall decreased DNA oxidation in both normal and stressed cells. It is suggested that GLS2’s control of ROS levels facilitates “the ability of p53 to protect cells from accumulation of genomic damage and allows cells to survive after mild and repairable genotoxic stress.”[4]
Structure
The structure of Glutaminase was determined using X-ray diffraction. The resolution of this protein is 1.73 Å. Resolution measures the quality of the data that has been collected on the crystal containing the protein. There are 2 chains containing 305 residues that make up the length of this dimeric protein. On each strand, 23% of Glutaminase, or 71 residues, are found in the 8 helices. Twenty-one percent, or 95 residues, construct the 23 beta sheet strands.[1]
Isozymes
Humans express the following two glutaminase isozymes:
|
| ||||||||||||||||||||||||||||||||||||||||
Related proteins
Glutaminases belong to a larger family that includes serine-dependent beta-lactamases and penicillin-binding proteins. Many bacteria have two isozymes. This model is based on selected known glutaminases and their homologs within prokaryotes, with the exclusion of highly-derived (long-branch) and architecturally varied homologs, so as to achieve conservative assignments. A sharp drop in scores occurs below 250, and cutoffs are set accordingly. The enzyme converts glutamine to glutamate, with the release of ammonia. Members tend to be described as glutaminase A (glsA), where B (glsB) is unknown and may not be homologous (as in Rhizobium etli; some species have two isozymes that may both be designated A (GlsA1 and GlsA2).
References
- ↑ 1.0 1.1 PDB 3A56; Hashizume R, Mizutani K, Takahashi N, Matsubara H, Matsunaga A, Yamaguchi S, Mikami B (2010). "Crystal structure of protein-glutaminase". to be published. doi:10.2210/pdb3a56/pdb.
- ↑ Olalla L, Gutiérrez A, Campos JA, Khan ZU, Alonso FJ, Segura JA, Márquez J, Aledo JC (Aug 2002). "Nuclear localization of L-type glutaminase in mammalian brain". J. Biol. Chem. 277 (41): 38939–38944. doi:10.1074/jbc.C200373200. PMID 12163477.
- ↑ Masola B, Ngubane NP (December 2010). "The activity of phosphate-dependent glutaminase from the rat small intestine is modulated by ADP and is dependent on integrity of mitochondria". Arch. Biochem. Biophys. 504 (2): 197–203. doi:10.1016/j.abb.2010.09.002. PMID 20831857.
- ↑ Suzuki S, Tanaka T, Poyurovsky MV, Nagano H, Mayama T, Ohkubo S, Lokshin M, Hosokawa H, Nakayama T, Suzuki Y, Sugano S, Sato E, Nagao T, Yokote K, Tatsuno I, Prives C (April 2010). "Phosphate-activated glutaminase (GLS2), a p53-inducible regulator of glutamine metabolism and reactive oxygen species". Proc. Natl. Acad. Sci. U.S.A. 107 (16): 7461–6. doi:10.1073/pnas.1002459107. PMC 2867754. PMID 20351271.
External links
- Glutaminase at the US National Library of Medicine Medical Subject Headings (MeSH)
| ||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||