Etretinate
 | |
|---|---|
| Systematic (IUPAC) name | |
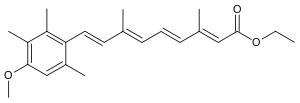
| ethyl 9-(4-methoxy-2,3,6-trimethyl-phenyl)- 3,7-dimethyl-nona- 2,4,6,8-tetraenoate | |
| Clinical data | |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| MedlinePlus | a601010 |
| Legal status | ? |
| Routes | Oral |
| Pharmacokinetic data | |
| Half-life | 120 days |
| Identifiers | |
| CAS number | 54350-48-0 |
| ATC code | D05BB01 |
| PubChem | CID 5282375 |
| DrugBank | DB00926 |
| ChemSpider | 4445538 |
| UNII | 65M2UDR9AG |
| KEGG | D00316 |
| ChEBI | CHEBI:4913 |
| ChEMBL | CHEMBL464 |
| Chemical data | |
| Formula | C23H30O3 |
| Mol. mass | 354.483 g/mol |
| SMILES
| |
| |
| | |
Etretinate (trade name Tegison) is a medication developed by Hoffmann–La Roche that was approved by the FDA in 1986 to treat severe psoriasis. It was subsequently removed from the Canadian market in 1996 and the United States market in 1998 due to the high risk of birth defects.
Properties
Etretinate has a low therapeutic index and a long elimination half-life (t1/2) of 120 days, which make dosing difficult.
Etretinate is an aromatic retinoid, and therefore highly lipophilic. It is stored and released from adipose tissue, so its effects can continue long after dosage stops. It is detectable in the plasma for up to three years following therapy.
Etretinate has been replaced by acitretin, a safer metabolite of etretinate.
Precautions
- Etretinate is a teratogen, and may cause birth defects long after use. Therefore, birth control is advised during therapy, and for at least three years after therapy has stopped.
- Etretinate should be avoided in children, as it may interfere with bone growth.
- If a patient has ever taken etretinate, he or she is not eligible to donate blood, due to the risk of birth defects.[1]
Side effects
- Hepatitis
- Pseudotumor cerebri
- Diffuse idiopathic skeletal Hyperostosis
References
- ↑ UK Blood Transfusion and Tissue Transplantation Services
| ||||||||||||||||||||||||||||||||