Chiraphos
| Chiraphos | |
|---|---|
 | |
| Other names * (2S,3S)-(–)-Bis(diphenylphosphino)butane
| |
| Identifiers | |
| CAS number | 74839-84-2 (R,R-Enantiomer) |
| PubChem | 10113249 |
| ChemSpider | 8288775 |
| Jmol-3D images | {{#if:P(c1ccccc1)(c2ccccc2)[C@H]([C@@H](P(c3ccccc3)c4ccccc4)C)C|Image 1 |
| |
| |
| Properties | |
| Molecular formula | C28H28P2 |
| Molar mass | 426.47 g/mol |
| Appearance | White powder |
| Melting point | 104–109 °C |
| Hazards | |
| EU classification | Irritant (XI) |
| R-phrases | R36/37/38 |
| S-phrases | S26 S37/39 |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| Infobox references | |
Chiraphos is a chiral diphosphine employed as a ligand in organometallic chemistry. This bidentate ligand chelates metals via the two phosphine groups. Its name is derived from its description — being both chiral and a phosphine. Chiraphos is available in two enantiomeric forms, S,S and R,R, each with C2 symmetry.
Preparation
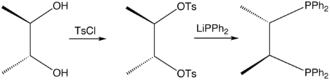
Chiraphos is prepared from S,S or R,R-2,3-butanediol, which are derived from commercially available S,S or R,R-tartaric acid; the technique of using cheaply available enantiopure starting materials is known as chiral pool synthesis. The diol is tosylated and then the ditosylate is treated with lithium diphenylphosphide.[1] The ligand was an important demonstration of how the conformation of the chelate ring can affect asymmetric induction by a metal catalyst. Prior to this work, in most chiral phosphines, e.g., DIPAMP, phosphorus was the stereogenic center.
References
- ↑ M. D. Fryzuk, B. Bosnich (1977). "Asymmetric synthesis. Production of optically active amino acids by catalytic hydrogenation". J. Am. Chem. Soc. 99 (19): 6262–6267. doi:10.1021/ja00461a014. PMID 893889.
- This article incorporates information from the revision as of 12:39, 21 June 2008 (UTC) of the equivalent article on the German Wikipedia.