Bepridil
From Wikipedia, the free encyclopedia
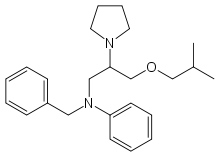 | |
|---|---|
| Systematic (IUPAC) name | |
| N-benzyl-N-(3-isobutoxy-2-pyrrolidin-1-yl-propyl)aniline | |
| Clinical data | |
| Trade names | Vascor |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a699051 |
| Pregnancy cat. | C (US) |
| Legal status | ? |
| Routes | Oral |
| Pharmacokinetic data | |
| Bioavailability | Well absorbed |
| Protein binding | 99% |
| Metabolism | Hepatic, CYP3A4-mediated |
| Half-life | 42 hours |
| Excretion | Renal |
| Identifiers | |
| CAS number | 64706-54-3 |
| ATC code | C08EA02 |
| PubChem | CID 2351 |
| IUPHAR ligand | 2337 |
| DrugBank | DB01244 |
| ChemSpider | 2261 |
| UNII | 755BO701MA |
| ChEBI | CHEBI:3061 |
| ChEMBL | CHEMBL1008 |
| Chemical data | |
| Formula | C24H34N2O |
| Mol. mass | 366.54 g/mol |
| SMILES
| |
| |
| | |
Bepridil (trade name Vascor) is a calcium channel blocker once used to treat angina. It is no longer sold in the United States.
It is nonselective.[1]
It has been discussed as a possible option in the treatment of atrial fibrillation.[2]
It has been implicated in causing ventricular arrhythmia (Torsade de pointes).
References
- ↑ Bezprozvanny I, Tsien RW (September 1995). "Voltage-dependent blockade of diverse types of voltage-gated Ca2+ channels expressed in Xenopus oocytes by the Ca2+ channel antagonist mibefradil (Ro 40-5967)". Mol. Pharmacol. 48 (3): 540–9. PMID 7565636.
- ↑ Imai S, Saito F, Takase H, et al. (May 2008). "Use of bepridil in combination with Ic antiarrhythmic agent in converting persistent atrial fibrillation to sinus rhythm". Circ. J. 72 (5): 709–15. doi:10.1253/circj.72.709. PMID 18441448.
External links
This article is issued from Wikipedia. The text is available under the Creative Commons Attribution/Share Alike; additional terms may apply for the media files.