2,3-Butanediol
| 2,3-Butanediol | |
|---|---|
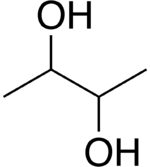 | |
| Butane-2,3-diol | |
| Other names 2,3-Butylene glycol | |
| Identifiers | |
| CAS number | 513-85-9 |
| PubChem | 262 |
| ChemSpider | 257 |
| EC number | 208-173-6 |
| ChEBI | CHEBI:62064 |
| Jmol-3D images | Image 1 |
| |
| |
| Properties | |
| Molecular formula | C4H10O2 |
| Molar mass | 90.121 g/mol |
| Appearance | nearly colorless solid or liquid |
| Odor | odorless |
| Density | 0.987 g/mL |
| Melting point | 19 °C; 66 °F; 292 K |
| Boiling point | 177 °C; 351 °F; 450 K |
| Solubility in water | miscible |
| Solubility | soluble in alcohol, ketones, ether |
| log P | -0.92 |
| Vapor pressure | 0.23 hPa (20 °C) |
| Acidity (pKa) | 14.9 |
| Refractive index (nD) | 1.4366 |
| Thermochemistry | |
| Std enthalpy of formation ΔfH |
-544.8 kJ/mol |
| Specific heat capacity, C | 213.0 J/K mol |
| Hazards | |
| EU classification | Flammable (F) |
| R-phrases | R11 |
| S-phrases | (S2) S7 S16 |
| NFPA 704 |
 1
1
0
|
| Flash point | 85 °C; 185 °F; 358 K |
| Autoignition temperature | 402 °C; 756 °F; 675 K |
| LD50 | 5462 mg/kg (rat, oral) |
| Related compounds | |
| Related butanediols | 1,4-Butanediol 1,3-Butanediol |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C (77 °F), 100 kPa) | |
| Infobox references | |
2,3-Butanediol is a chemical compound composed of carbon, hydrogen, and oxygen. Its formula is C4H10O2. It is one of the constitutional isomers of butanediol.[1]
Uses and manufacturing
The (2R,3R)-stereoisomer of 2,3-butanediol is produced by a variety of microorganisms in a process known as butanediol fermentation. It is found naturally in cocoa butter, in the roots of Ruta graveolens, sweet corn, and in rotten mussels. It is used in the resolution of carbonyl compounds in gas chromatography.[2]
History
During World War II research was done towards producing 2,3-butanediol by fermentation in order to produce 1,3-butadiene, the monomer of the polybutadiene used in a leading type of synthetic rubber.[3]
References
- ↑ 2,3-Butanediol at SigmaAldrich
- ↑ "3,5-dinitrobenzoic acid". Combined Chemical Dictionary. Chapman and Hall/CRC Press. 2007.
- ↑ "Fermentation Derived 2,3-Butanediol", by Marcio Voloch et al. in Comprehensive Biotechnology, Pergamon Press Ltd., England Vol 2, Section 3, p. 933 (1986).