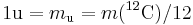Atomic mass unit
|
|
|
| Unit system: | SI recognized unit |
| Unit of... | mass |
| Symbol: | u |
|
|
|
| 1 u in... | is equal to... |
| dalton | 1 |
| kg | 1.660538921(73)×10−27 |
| eV/c2 | 931.494061(21)×106 |
| me | 1822.88839 |
The unified atomic mass unit (Also known as amu) (symbol: u) or dalton (symbol: Da) is a unit that is used for indicating mass on an atomic or molecular scale. It is defined as one twelfth of the rest mass of an unbound neutral atom of carbon-12 in its nuclear and electronic ground state,[1] and has a value of 1.660538921(73)×10−27 kg.[2] One dalton is approximately equal to the mass of one proton or one neutron. The CIPM have categorised it as a "non-SI unit whose values in SI units must be obtained experimentally".[1]
Contents[hide] |
History
The atomic weight scale has traditionally been a relative scale, that is without an explicit unit, with the first atomic weight basis suggested by John Dalton in 1803 as 1H.[3] Despite the initial mass of 1H being used as the natural unit for atomic weight, it was suggested by Wilhelm Ostwald that atomic weights would be best expressed in terms in units of 1/16 weight of oxygen. This evaluation was made prior to the discovery of the existence of elemental isotopes, which occurred in 1912.[3]
The discovery of isotopic oxygen in 1929 led to a divergence in atomic weight representation, with isotopically weighted oxygen (chemistry) and pure 16O (physics) bases both used as the basis for the atomic mass unit (amu). The inevitable divergence could result in errors in computations, and was thus unwieldy. The reference was changed to carbon-12 in 1961[4] and a new symbol "u" replaced the now deprecated "amu".
The current unit is referred to as the "unified atomic mass unit" u.[5] The choice of carbon-12 was used to minimise further divergence with prior literature.[3]
Terminology
The unified atomic mass unit and the dalton are different names for the same unit of measure. Since the dalton was first introduced, there has been a gradual change towards using it in preference to the unified atomic mass unit.
- In 1993, the International Union of Pure and Applied Chemistry approved the use of the dalton with the qualification that the GCPM had not given its approval.[6]
- In 2003 the Consultative Committee for Units, part of the CIPM, recommended a preference for the usage of the "dalton" over the "unified atomic mass unit" as it "is shorter and works better with prefixes".[7]
- In 2005, the International Union of Pure and Applied Physics endorsed the use of the dalton as an alternative to the unified atomic mass unit.[8]
- In 2006, in the 8th edition of the formal definition of SI, the CIPM cataloged the dalton alongside the unified atomic mass unit as a "Non-SI units whose values in SI units must be obtained experimentally: Units accepted for use with the SI".[1] The definition also noted that "The dalton is often combined with SI prefixes ..."
- In 2009, when the International Organization for Standardization published updated versions of ISO 80000, it gave mixed messages as to whether or not the unified atomic mass unit had been deprecated: ISO ISO 80000-1:2009 (General), identified the dalton as having "earlier [been] called the unified atomic mass unit u",[9] but ISO 80000-10:2009 (atomic and nuclear physics) catalogued both as being alternatives for each other.[10]
- The 2010 version of the Oxford University Press style guide for authors in life sciences gave the following guidance "Use the Système international d'unités (SI) wherever possible ... The Dalton (Da) or more conveniently the kDa is a permitted non-SI unit for molecular mass or mass of a particular band in a separating gel."[11] At the same time, the author guidelines for the journal "Rapid Communications in Mass Spectrometry" stated "The Dalton (Da) is a unit of mass normally used for the molecular weight ... use of the Da in place of the u has become commonplace in the mass spectrometry literature ... The "atomic mass unit", abbreviated "amu", is an archaic unit".[12]
Relationship to SI
The definition of the mole, an SI base unit, was accepted by the CGPM in 1971 as:
- The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; its symbol is "mol".
- When the mole is used, the elementary entities must be specified and may be atoms, molecules, ions, electrons, other particles, or specified groups of such particles.
The definition of the mole also determines the value of the universal constant that relates the number of entities to amount of substance for any sample. This constant is called the Avogadro constant, symbol NA or L, and is equal to 6.02214129(27)×1023 entities per mole.[13]
Given that the unified atomic mass unit is one twelfth the mass of one atom of carbon 12, meaning the mass of such an atom is 12u, it follows that there are NA atoms of carbon 12 in 0.012 kg of carbon 12. This can be expressed mathematically as
- NA(12u) = 0.012 kg/mol, or
- NAu = 0.001 kg/mol
See also
References
- ^ a b c International Bureau of Weights and Measures (2006), The International System of Units (SI) (8th ed.), p. 126, ISBN 92-822-2213-6, http://www.bipm.org/utils/common/pdf/si_brochure_8_en.pdf
- ^ Fundamental Physical Constants from NIST
- ^ a b c Petley, B. W., "The atomic mass unit", IEEE Trans. Instrum. Meas. 38 (2): 175–79, doi:10.1109/19.192268
- ^ Holden, Norman E. (2004), "Atomic Weights and the International Committee—A Historical Review", Chem. Int. 26 (1): 4–7, http://www.iupac.org/publications/ci/2004/2601/1_holden.html
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "unified atomic mass unit".
- ^ Mills, Ian; Cvitaš, Tomislav; Homann, Klaus; Kallay, Nikola; Kuchitsu, Kozo (1993). Quantities, Units and Symbols in Physical Chemistry International Union of Pure and Applied Chemistry; Physical Chemistry Division (2nd ed.). International Union of Pure and Applied Chemistry and published for them by Blackwell Science Ltd. ISBN 0-632-03583-8. http://www.iupac.org/publications/books/gbook/green_book_2ed.pdf.
- ^ "Consultative Committee for Units (CCU); Report of the 15th meeting (17 –18 April 2003) to the International Committee for Weights and Measures". http://www.bipm.org/utils/common/pdf/CCU15.pdf. Retrieved 14th Aug 2010.
- ^ "IU14. IUPAC Interdivisional Committee on Nomenclature and Symbols (ICTNS)". http://www.iupap.org/commissions/interunion/iu14/ga-05.html. Retrieved 2010-08-14.
- ^ International Standard ISO 80000-1:2009 – Quantities and Units – Part 1: General, International Organization for Standardization, 2009
- ^ International Standard ISO 80000-10:2009 – Quantities and units – Part 10: Atomic and nuclear physics, International Organization for Standardization, 2009
- ^ "Instructions to Authors". AoB Plants. Oxford journals; Oxford University Press. http://www.oxfordjournals.org/our_journals/aobpla/for_authors/. Retrieved 2010-08-22.
- ^ "Author guidelines". Rapid Communications in Mass Spectrometry (Wiley-Blackwell). 2010. http://en.wikipedia.org/w/index.php?title=Atomic_mass_unit&action=edit§ion=2. Retrieved 2011-05-08.
- ^ International Bureau of Weights and Measures (2006), The International System of Units (SI) (8th ed.), pp. 114,115, ISBN 92-822-2213-6, http://www.bipm.org/utils/common/pdf/si_brochure_8_en.pdf
External links
- atomic mass unit at sizes.com
|
||||||||||||||||||||