Centrifugation
Centrifugation is a process that involves the use of the centrifugal force for the sedimentation of mixtures with a centrifuge, used in industry and in laboratory settings. More-dense components of the mixture migrate away from the axis of the centrifuge, while less-dense components of the mixture migrate towards the axis. Chemists and biologists may increase the effective gravitational force on a test tube so as to more rapidly and completely cause the precipitate ("pellet") to gather on the bottom of the tube. The remaining solution is properly called the "supernate" or "supernatant liquid". The supernatant liquid is then either quickly decanted from the tube without disturbing the precipitate, or withdrawn with a Pasteur pipette.
The rate of centrifugation is specified by the angular velocity measured in revolutions per minute (RPM), or acceleration expressed as g. The conversion factor between RPM and g depends on the radius of the sample in the centrifuge rotor. The particles' settling velocity in centrifugation is a function of their size and shape, centrifugal acceleration, the volume fraction of solids present, the density difference between the particle and the liquid, and the viscosity.
In the chemical and food industries, special centrifuges can process a continuous stream of particle-laden liquid.
Centrifugation is the most common method used for uranium enrichment, relying on the slight mass difference between atoms of U238 and U235 in uranium hexafluoride gas.
Contents |
Centrifugation in biological research
Microcentrifuges
Microcentrifuges are used to process small volumes of biological molecules, cells, or nuclei. Microcentrifuge tubes generally hold 0.5 - 2 mL of liquid, and are spun at maximum angular speeds of 12000–13000 rpm. Microcentrifuges are small enough to fit on a table-top and have rotors that can quickly change speeds. They may or may not have a refrigeration function. This is the important process.
High-speed centrifuges
High-speed or superspeed centrifuges can handle larger sample volumes, from a few tens of millilitres to several litres. Additionally, larger centrifuges can also reach higher angular velocities (around 30000 rpm). The rotors may come with different adapters to hold various sizes of test tubes, bottles, or microtiter plates.
Fractionation Process
Method of fractionation procedure: Cell sample is stored in a suspension which is:
- Buffered - neutral Ph, preventing damage to the structure of proteins including enzymes (which could affect ionic bonds)
- Isotonic (of equal water potential) - this prevents water gain or loss by the organelles
- Cool - reducing the overall activity of enzyme released later in the procedure
- Cells are homogenised in a blender and filtered to remove debris
- The homogenised sample is placed in an ultracentrifuge and spun in low speed - nuclei settle out, forming a pellet
- The supernatant (suspension containing remaining organelles) is spun at a higher speed - chloroplasts settle out
- The supernatant is spun at a higher speed still - mitochondria and lysosomes settle out
- The supernatant is spun at an even higher speed - ribosomes, membranes settle out
The ribosomes, membranes and Golgi complexes can be separated by another technique called density gradient centrifugation.
Ultracentrifuges
Ultracentrifugation makes use of high centrifugal force for studying properties of biological particles. Compared to microcentrifuges or high-speed centrifuges, ultracentrifuges can isolate much smaller particles, including ribosomes, proteins, and viruses. Ultracentrifuges can also be used in the study of membrane fractionation. This occurs because ultracentrifuges can reach maximum angular velocities in excess of 70000 rpm. Additionally, while microcentrifuges and supercentrifuges separate particles in batches (limited volumes of samples must be handled manually in test tubes or bottles), ultracentrifuges can separate molecules in batch or continuous flow systems.
In addition to purification, analytical ultracentrifugation (AUC) can be used for determination of the properties of macromolecules such as shape, mass, composition, and conformation. Samples are centrifuged with a high-density solution such as sucrose, caesium chloride, or iodixanol. The high-density solution may be at a uniform concentration throughout the test tube ("cushion") or a varying concentration ("gradient"). Molecular properties can be modeled through sedimentation velocity analysis or sedimentation equilibrium analysis. During the run, the particles or molecules will migrate through the test tube at different speeds depending on their physical properties and the properties of the solution, and eventually form a pellet at the bottom of the tube, or bands at various heights.
Centrifugation analysis
Lamm equation
Particle dispersion and sedimentation can be analyzed using the Lamm equation. The calculation of the sedimentation coefficient and diffusion coefficient is useful for determining the physical properties of the molecule, including shape and conformational changes. However, the Lamm equation is most ideal for modeling concentrations of ideal, non-interacting solutes. Chemical reactions are unaccounted for by this equation. Additionally, for large molecular weight particles, sedimentation is not always smooth. This may lead to the overestimation of the diffusion coefficient, or oscillation effects at the bottom of a solution cell.
Sigma analysis
Sigma analysis is a useful tool determining centrifuge properties. It is similar to the continuity equation that relates volumetric flow rate Q, fluid velocity u, and flow path cross-sectional Area A:
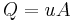
In the case of sigma analysis, u is replaced by vg,the settling velocity at centripetal acceleration of g (9.81 m/s2), Σ replaces area, and is a property of the type of centrifuge, and Q is the input fluid flow rate. Σ has the same units as area.
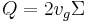
Other applications
- Separating textile.
- Removing water from lettuce after washing it in a salad spinner
- Separating particles from an air-flow using cyclonic separation.
- The clarification and stabilization of wine
- It is used in washing machines to separate water particles from clothes while drying in washing machines
Sources
- Harrison, Roger G., Todd, Paul, Rudge, Scott R., Petrides D.P. Bioseparations Science and Engineering. Oxford University Press, 2003.
- Dishon, M., Weiss, G.H., Yphantis, D.A. Numerical Solutions of the Lamm Equation. I. Numerical Procedure. Biopolymers, Vol. 4, 1966. pp. 449–455.
- Cao, W., Demeler B. Modeling Analytical Ultracentrifugation Experiments with an Adaptive Space-Time Finite Element Solution for Multicomponent Reacting Systems. Biophysical Journal, Vol. 95, 2008. pp. 54–65.
- Cole, J.L., Hansen, J.C. Analytical Ultracentrifugation as a Contemporary Biomolecular Research Tool. Methods and Reviews, 1999/2000.
- Howlett, G.J., Minton, A.P., Rivas, G. Analytical Ultracentrifugation for the Study of Protein Association and Assembly. Current Opinion in Chemical Biology, Vol. 10, 2006. pp. 430–436.
- Dam, J., Velikovsky, C.A., Mariuzza R.A., et al. Sedimentation Velocity Analysis of Heterogeneous Protein-Protein Interactions: Lamm Equation Modeling and Sedimentation Coefficient Distributions c(s). Biophysical Journal, Vol. 89, 2005. pp. 619–634.
- Berkowitz, S.A., Philo, J.S. Monitoring the Homogeneity of Adenovirus Preparations (a Gene Therapy Delivery System) Using Analytical Ultracentrifugation. Analytical Biochemistry, Vol. 362, 2007. pp. 16–37.