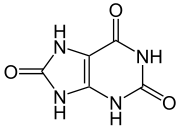
Uric acid
| Uric acid | |
|---|---|
 |
 |
|
7,9-dihydro-1H-purine-
2,6,8(3H)-trione |
|
|
Other names
2,6,8 Trioxypurine
|
|
| Identifiers | |
| CAS number | 69-93-2 |
| PubChem | 1175 |
| ChemSpider | 1142 |
| EC number | 200-720-7 |
| KEGG | C00366 |
|
SMILES
C12NC(=O)NC(=O)C=2NC(=O)N1
|
|
|
InChI
InChI=1/C5H4N4O3/c10-3-1-2(7-4(11)6-1)8-5(12)9-3/h(H4,6,7,8,9,10,11,12)/f/h6-9H[1]
|
|
| Properties | |
| Molecular formula | C5H4N4O3 |
| Molar mass | 168g/mol |
| Appearance | White Crystals |
| Density | 1.87 |
| Melting point |
decomposes on heating |
| Boiling point |
N/A |
| Solubility in water | Slightly |
| Acidity (pKa) | 5.8 |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) |
|
| Infobox references | |
Uric acid (or urate) is a heterocyclic compound of carbon, nitrogen, oxygen, and hydrogen with the formula C5H4N4O3.
Contents |
Chemistry
Uric acid is a diprotic acid with pKa1=5.4 and pKa2=10.3[2]. Thus in strong alkali at high pH it forms the dually charged full urate ion, but at biological pH or in the presence of carbonic acid or carbonate ions it forms the singly charged hydrogen or acid urate ion as its pKa2 is greater than the pKa1 of carbonic acid. As its second ionization is so weak the full urate salts tend to hydrolyse back to hydrogen urate salts and free base at pH values around neutral. It is aromatic because of the purine functional group.
As a bicyclic, heterocyclic purine derivative, uric acid does not protonate in the same manner as do carboxylic acids. X-Ray diffraction studies on the hydrogen urate ion in crystals of ammomium hydrogen urate, formed in vivo as gouty deposits, revealed that the keto-oxygen in the 2 position of a tautomer of the purine structure existed as a hydroxyl group and that the two flanking nitrogen atoms at the 1 and 3 positions shared the ionic charge in the six membered pi-resonance-stabilized ring.[3]
Thus, whereas most organic acids are deprotonated by the ionization of a polar hydrogen-to-oxygen bond, usually accompanied by some form of resonance stabilization (resulting in a carboxylate ion), this acid is deprotonated at a nitrogen atom and uses a tautomeric keto/hydroxy group as an electron-withdrawing group to increase the pK1 value. The five membered ring also possesses a keto group (in the 8 position), flanked by two secondary amino groups (in the 7 and 9 positions), and deprotonation of one of these at high pH could explain the pK2 and behavior as a diprotic acid. Similar tautomeric rearrangement and pi-resonance stabilization would then give the ion some degree of stability. (On the structure shown at the upper right, the NH at the upper right on the six membered ring is "1", counting clockwise around the six membered ring to "6" for the keto carbon at the top of the six membered ring. The upper most NH on the five membered ring is "7", counting counter clockwise around this ring to the lower NH, which is "9".)
Solubility of uric acid and its salts
Generally the solubilities of uric acid, its Alkali and Alkali Earth Metal salts in water are rather low and all exhibit greater solubility in hot water than cold allowing for easy recrystallization. The solubility of the acid and its salts in ethanol is very low or negligible. In ethanol water mixtures the solubilities are somewhere between the end values for pure ethanol and pure water.
| Compound | Cold Water | Boiling Water |
|---|---|---|
| Uric Acid | 15000 | 2000 |
| NH4HUrate | - | 1600 |
| LiHUrate | 370 | 39 |
| NaHUrate | 1175 | 124 |
| KHUrate | 790 | 75 |
| Mg(HUrate)2 | 3750 | 160 |
| Ca(HUrate)2 | 603 | 276 |
| Na2Urate | 77 | - |
| K2Urate | 44 | 35 |
| CaUrate | 1500 | 1440 |
| SrUrate | 4300 | 1790 |
| BaUrate | 7900 | 2700 |
The figures given indicate what mass of water is required to dissolve a unit mass of compound indicated, the lower the number, the more soluble the substance in the said solvent.[4][5][6]
Biology
Uric acid is produced by xanthine oxidase from xanthine and hypoxanthine, which in turn are produced from purine. Uric acid is more toxic to tissues than either xanthine or hypoxanthine. Uric acid is released in hypoxic conditions.[7]
In humans and higher primates, uric acid is the final oxidation (breakdown) product of purine metabolism and is excreted in urine. In most other mammals, the enzyme uricase further oxidizes uric acid to allantoin.[8] The loss of uricase in higher primates parallels the similar loss of the ability to synthesize ascorbic acid.[9] Both uric acid and ascorbic acid are strong reducing agents (electron donors) and potent antioxidants. In humans, over half the antioxidant capacity of blood plasma comes from uric acid.[10] The Dalmatian dog has a genetic defect in uric acid uptake by the liver, resulting in decreased conversion to allantoin, so this breed excretes uric acid, and not allantoin, in the urine.[11]
In birds and reptiles, and in some desert dwelling mammals (e.g., the kangaroo rat), uric acid also is the end product of purine metabolism, but it is excreted in feces as a dry mass. This involves a complex metabolic pathway that is energetically costly in comparison to processing of other nitrogenous wastes such as urea (from urea cycle) or ammonia, but has the advantage of reducing water loss.[12]
In humans, about 70% of daily uric acid disposal occurs via the kidneys, and in 5-25% of humans impaired renal (kidney) excretion leads to hyperuricemia.[13]
Genetics
A proportion of people have mutations in the proteins responsible for the excretion of uric acid by the kidneys. Nine genes have so far been identified: SLC2A9; ABCG2; SLC17A1; SLC22A11; SLC22A12; SLC16A9; GCKR; LRRC16A; and PDZK1.[14][15] SLC2A9 is known to transport both uric acid and fructose.[13][16]
Medicine
In human blood plasma, the reference range of uric acid is between 3.6 mg/dL (~214µmol/L) and 8.3 mg/dL (~494µmol/L) (1 mg/dL=59.48 µmol/L).[17] This range is considered normal by the American Medical Association. Uric acid concentrations in blood plasma above and below the normal range are known, respectively, as hyperuricemia and hypouricemia. Similarly, uric acid concentrations in urine above and below normal are known as hyperuricosuria and hypouricosuria. Such abnormal concentrations of uric acid are not medical conditions, but are associated with a variety of medical conditions.

High uric acid
Gout
Excess serum accumulation of uric acid can lead to a type of arthritis known as gout.[18] This painful condition is the result of needle-like crystals of uric acid precipitating in joints and capillaries.
Elevated serum uric acid (hyperuricemia) can result from high intake of purine-rich foods, and/or impaired excretion by the kidneys. Saturation levels of uric acid in blood may result in one form of kidney stones when the urate crystallizes in the kidney. These uric acid stones are radiolucent and so do not appear on an abdominal plain x-ray or CT scan. Their presence must be diagnosed by ultrasound for this reason. Very large stones may be detected on x-ray by their displacement of the surrounding kidney tissues. Some patients with gout eventually get uric kidney stones.
Gout can occur where serum uric acid levels are as low as 6 mg/dL (~357µmol/L), but an individual can have serum values as high as 9.6 mg/dL (~565µmol/L) and not have gout.[19]
One treatment for gout has been administration of Lithium salts; lithium urate is more soluble.
Lesch-Nyhan syndrome
Lesch-Nyhan syndrome, an extremely rare inherited disorder, is also associated with very high serum uric acid levels.[20]
Spasticity, involuntary movement and cognitive retardation as well as manifestations of gout are seen in cases of this syndrome.[21]
Cardiovascular disease
Although uric acid can act as an antioxidant, excess serum accumulation is often associated with cardiovascular disease. It is not known whether this is causative (e.g., by acting as a prooxidant ) or a protective reaction taking advantage of urate's antioxidant properties.[18]
Diabetes
The association of high serum uric acid with insulin resistance has been known since the early part of the 20th century, nevertheless, recognition of high serum uric acid as a risk factor for diabetes has been a matter of debate. In fact, hyperuricemia has always been presumed to be a consequence of insulin resistance rather than its precursor.[22] However, it was shown in a prospective follow-up study that high serum uric acid is associated with higher risk of type 2 diabetes independent of obesity, dyslipidemia, and hypertension.[23]
Metabolic syndrome
Hyperuricemia is associated with components of metabolic syndrome and it has been debated for a while to be a component of it. It has been shown in a recent study that fructose-induced hyperuricemia may play a pathogenic role in the metabolic syndrome. This is consistent with the increased consumption in recent decades of fructose-containing beverages (such as fruit juices and soft drinks sweetened with sugar and high-fructose corn syrup) and the epidemic of diabetes and obesity.[24]
Uric acid stone formation
Uric acid stones, which form in the absence of secondary causes such as chronic diarrhea, vigorous exercise, dehydration, and animal protein loading, are felt to be secondary to obesity and insulin resistance seen in metabolic syndrome. Increased dietary acid leads to increased endogenous acid production in the liver and muscles which in turn leads to an increased acid load to the kidneys. This load is handled more poorly because of renal fat infiltration and insulin resistance which are felt to impair ammonia excretion (a buffer). The urine is therefore quite acidic and uric acid becomes insoluble, crystallizes and stones form. In addition, naturally present promotor and inhibitor factors may be affected. This explains the high prevalence of uric stones and unusually acidic urine seen in patients with type 2 diabetes. Uric acid crystals can also promote the formation of calcium oxalate stones, acting as "seed crystals" (heterogeneous nucleation).[25]
Causes of high uric acid
- In many instances, people have elevated uric acid levels for hereditary reasons.
- Diet may be a factor: as mentioned above in Metabolic Syndrome, fructose (and sucrose) can cause increased levels of uric acid. Eating large amounts of sea salt can cause increased levels of uric acid. (Medical consultation is recommended before using large quantities of sea salt in daily cooking.)
- Serum uric acid can be elevated due to reduced excretion by the kidneys
- Serum uric acid can be elevated due to high intake of dietary purine.
- Fe activates xanthine oxidase (XO) and Cu deactivates it, so that as men accumulate Fe with age (ferritin levels rise above 45 ng/dl) and Cu levels decline as testosterone levels drop with age (testosterone increases Cu half life), eventually the high Fe/Cu results in more active XO and higher urate levels. Excess Fe can be eliminated through phlebotomy (blood donation) and low Cu can be corrected through daily intake of 2 mg Cu per day, reducing urate levels.
Low uric acid
Multiple sclerosis
Lower serum values of uric acid have been associated with Multiple Sclerosis. Multiple sclerosis (MS) patients have been found to have serum levels ~194µmol/L, with patients in relapse averaging ~160µmol/L and patients in remission averaging ~230µmol/L. Serum uric acid in healthy controls was ~290µmol/L.[26] Conversion factor: 1 mg/dL=59.48 µmol/L[17]
A 1998 study completed a statistical analysis of 20 million patient records, comparing serum uric acid values in patients with gout and patients with multiple sclerosis. Almost no overlap between the groups was found.[27]
Uric acid has been successfully used in the treatment and prevention of the animal (murine) model of MS. A 2006 study found that elevation of serum uric acid values in multiple sclerosis patients, by oral supplementation with inosine, resulted in lower relapse rates, and no adverse effects.[28]
Causes of low uric acid
Low uric acid (hypouricemia) can have numerous causes.
Low dietary zinc intakes cause lower uric acid levels. This effect can be even more pronounced in women taking oral contraceptive medication.[29]
Xanthine oxidase is an Fe-Mo enzyme, so people with Fe deficiency (the most cammon cause of anemia in young women) or Mo deficiency can experience hypouricemia.
Xanthine oxidase loses its function and gains ascorbase function when some of the Fe atoms in Xanthine oxidase are replaced with Cu atoms. Accordingly, people with high Cu/Fe can experience hypouricemia anc vitamin C deficiency, resulting in oxidative damage. Since estrogen increases the half life of Cu, women with very high estrogen levels and intense blood los during menstruation are likely to have a high Cu/Fe and present with hypouricemia.
Sevelamer, a drug indicated for prevention of hyperphosphataemia in patients with chronic renal failure, can significantly reduce serum uric acid.[30]
Normalizing low uric acid
Correcting low or deficient zinc levels can help elevate serum uric acid.[31] Inosine can be used to elevate uric acid levels.[26] Zn inhibits Cu absorption, helping to reduce the high Cu/Fe in some people with hypouricemia. Fe supplements can ensure adequate Fe reserves (ferritin above 25 ng/dl), also correcting the high Cu/Fe.
Oxidative stress
Uric acid may be a marker of oxidative stress,[32] and may have a potential therapeutic role as an antioxidant.[33] On the other hand, like other strong reducing substances such as ascorbate, uric acid can also act as a prooxidant,[34] particularly at elevated levels. Thus, it is unclear whether elevated levels of uric acid in diseases associated with oxidative stress such as stroke and atherosclerosis are a protective response or a primary cause.[35][36]
For example, some researchers propose that hyperuricemia-induced oxidative stress is a cause of metabolic syndrome.[24][37] On the other hand, plasma uric acid levels correlate with longevity in primates and other mammals.[38] This is presumably a function of urate's antioxidant properties.
Sources of uric acid
- In humans Purines are excreted as uric acid. Purines are found in high amounts in animal food products, such as liver and sardines.[39] A moderate amount of purine is also contained in beef, pork, poultry, fish and seafood, asparagus, cauliflower, spinach, mushrooms, green peas, lentils, dried peas, beans, oatmeal, wheat bran and wheat germ.[40]
- Examples of high purine and Fe sources include: sweetbreads, anchovies, sardines, liver, beef kidneys, brains, meat extracts (e.g., Oxo, Bovril), herring, mackerel, scallops, game meats, and gravy.
- Moderate intake of purine-containing food is not associated with an increased risk of gout.[41]
Other uric acid facts
The high nitrogen content of uric acid makes guano a useful agricultural fertilizer.
The crystalline form of uric acid is used as a reflector in certain species of fireflies.
The uric acid in urine can also dry in a baby's diaper to form a pinkish powder that is harmless.
Urate is being researched for its possible antioxidant properties in treating Parkinson's disease.[42]
See also
- Hyperuricemia
References
- ↑ "Uric Acid." Biological Magnetic Resonance Data Bank. Indicator Information Retrieved on 18 February 2008.
- ↑ Uric Acid, Francis H. McCrudden
- ↑ European Powder Diffraction Conference, EPDIC-9
- ↑ C.R.C. 62nd Ed.
- ↑ MERK Index, Ninth Ed.
- ↑ Uric Acid, Francis H. McCrudden, page 58
- ↑ Baillie, J.K.; M.G. Bates, A.A. Thompson, W.S. Waring, R.W. Partridge, M.F. Schnopp, A. Simpson, F. Gulliver-Sloan, S.R. Maxwell, D.J. Webb (2007-05). "Endogenous urate production augments plasma antioxidant capacity in healthy lowland subjects exposed to high altitude". Chest 131 (5): 1473–1478. doi:10.1378/chest.06-2235. PMID 17494796.
- ↑ Angstadt, Carol N. (1997-12-04). Purine and Pyrimidine Metabolism: Purine Catabolism. NetBiochem, 4 December 1997. Retrieved from http://library.med.utah.edu/NetBiochem/pupyr/pp.htm#Pu%20Catab.
- ↑ Proctor P (1970-11). "Similar functions of uric acid and ascorbate in man?". Nature 228 (5274): 868. doi:10.1038/228868a0. PMID 5477017.
- ↑ S. R. J. Maxwell, H. Thomason, D. Sandler, C. Leguen, M. A. Baxter, G. H. G. Thorpe, A. F. Jones, A. H. Barnett. "Antioxidant status in patients with uncomplicated insulin-dependent and non-insulin-dependent diabetes mellitus" European Journal of Clinical Investigation (1997) 27, 484-490. Blackwell Science Ltd., retrieved from http://www3.interscience.wiley.com/journal/119145773/abstract
- ↑ Friedman, Meyer; and Byers, Sanford O. (1 September 1948). "Observations concerning the causes of the excess excretion of uric acid in the Dalmatian dog". The Journal of Biological Chemistry 175 (2): 727–35. PMID 18880769. http://www.jbc.org/cgi/reprint/175/2/727.
- ↑ Hazard, Lisa C. (2004). Sodium and Potassium Secretion by Iguana Salt Glands. University of California Press. pp. 84–85. ISBN 9780520238541.
- ↑ 13.0 13.1 Vitart V, Rudan I, Hayward C, et al. (2008-04). "SLC2A9 is a newly identified urate transporter influencing serum urate concentration, urate excretion and gout". Nature Genetics 40 (4): 437–42. doi:10.1038/ng.106. PMID 18327257.
- ↑ Aringer M, Graessler J (December 2008). "Understanding deficient elimination of uric acid". Lancet 372 (9654): 1929–30. doi:10.1016/S0140-6736(08)61344-6. PMID 18834627.
- ↑ Kolz M, Johnson T , et al. (June 2009). "Meta-analysis of 28,141 individuals identifies common variants within five new loci that influence uric acid concentrations". PLoS Genet 5 (6): e1000504. doi:10.1371/journal.pgen.1000504. PMID 19503597.
- ↑ Döring A, Gieger C, Mehta D, et al. (April 2008). "SLC2A9 influences uric acid concentrations with pronounced sex-specific effects". Nature Genetics 40 (4): 430–6. doi:10.1038/ng.107. PMID 18327256.
- ↑ 17.0 17.1 SI Units for Clinical Data
- ↑ 18.0 18.1 Heinig M, Johnson RJ (December 2006). "Role of uric acid in hypertension, renal disease, and metabolic syndrome". Cleveland Clinic Journal of Medicine 73 (12): 1059–64. doi:10.3949/ccjm.73.12.1059. PMID 17190309.
- ↑ Tausche AK, Unger S, Richter K, et al. (May 2006). "Hyperurikämie und Gicht [Hyperuricemia and gout: diagnosis and therapy]" (in German). Der Internist 47 (5): 509–20; quiz 521. doi:10.1007/s00108-006-1578-y. PMID 16586130.
- ↑ Luo YC, Do JS, Liu CC (October 2006). "An amperometric uric acid biosensor based on modified Ir-C electrode". Biosensors & Bioelectronics 22 (4): 482–8. doi:10.1016/j.bios.2006.07.013. PMID 16908130.
- ↑ Nyhan WL (March 2005). "Lesch-Nyhan Disease". Journal of the History of the Neurosciences 14 (1): 1–10. doi:10.1080/096470490512490. PMID 15804753.
- ↑ Cappuccio FP, Strazzullo P, Farinaro E, Trevisan M (July 1993). "Uric acid metabolism and tubular sodium handling. Results from a population-based study". JAMA 270 (3): 354–9. doi:10.1001/jama.270.3.354. PMID 8315780.
- ↑ Dehghan A, van Hoek M, Sijbrands EJ, Hofman A, Witteman JC (February 2008). "High serum uric acid as a novel risk factor for type 2 diabetes". Diabetes Care 31 (2): 361–2. doi:10.2337/dc07-1276. PMID 17977935.
- ↑ 24.0 24.1 Nakagawa T, Hu H, Zharikov S, et al. (March 2006). "A causal role for uric acid in fructose-induced metabolic syndrome". American Journal of Physiology. Renal Physiology 290 (3): F625–31. doi:10.1152/ajprenal.00140.2005. PMID 16234313.
- ↑ Pak CY (September 2008). "Medical stone management: 35 years of advances". The Journal of Urology 180 (3): 813–9. doi:10.1016/j.juro.2008.05.048. PMID 18635234.
- ↑ 26.0 26.1 Toncev G, Milicic B, Toncev S, Samardzic G (May 2002). "Serum uric acid levels in multiple sclerosis patients correlate with activity of disease and blood-brain barrier dysfunction". European Journal of Neurology 9 (3): 221–6. doi:10.1046/j.1468-1331.2002.00384.x. PMID 11985629.
- ↑ Hooper DC, Spitsin S, Kean RB, et al. (January 1998). "Uric acid, a natural scavenger of peroxynitrite, in experimental allergic encephalomyelitis and multiple sclerosis". Proceedings of the National Academy of Sciences of the United States of America 95 (2): 675–80. doi:10.1073/pnas.95.2.675. PMID 9435251.
- ↑ Toncev G (October 2006). "Therapeutic value of serum uric acid levels increasing in the treatment of multiple sclerosis". Vojnosanitetski Pregled 63 (10): 879–82. PMID 17121380.
- ↑ Hess FM, King JC, Margen S (1 December 1977). "Effect of low zinc intake and oral contraceptive agents on nitrogen utilization and clinical findings in young women". The Journal of Nutrition 107 (12): 2219–27. PMID 925768. http://jn.nutrition.org/cgi/pmidlookup?view=long&pmid=925768.
- ↑ Garg JP, Chasan-Taber S, Blair A, et al. (January 2005). "Effects of sevelamer and calcium-based phosphate binders on uric acid concentrations in patients undergoing hemodialysis: a randomized clinical trial". Arthritis and Rheumatism 52 (1): 290–5. doi:10.1002/art.20781. PMID 15641045.
- ↑ Umeki S, Ohga R, Konishi Y, Yasuda T, Morimoto K, Terao A (November 1986). "Oral zinc therapy normalizes serum uric acid level in Wilson's disease patients". The American Journal of the Medical Sciences 292 (5): 289–92. doi:10.1097/00000441-198611000-00007. PMID 3777013.
- ↑ Becker BF (June 1993). "Towards the physiological function of uric acid". Free Radical Biology & Medicine 14 (6): 615–31. doi:10.1016/0891-5849(93)90143-I. PMID 8325534.
- ↑ Glantzounis GK, Tsimoyiannis EC, Kappas AM, Galaris DA (2005). "Uric acid and oxidative stress". Current Pharmaceutical Design 11 (32): 4145–51. doi:10.2174/138161205774913255. PMID 16375736.
- ↑ Proctor P (1972). "Electron-transfer factors in psychosis and dyskinesia". Physiological Chemistry and Physics 4 (4): 349–60. PMID 4680784.
- ↑ Free Radicals and Human Disease
- ↑ Proctor PH (May 2008). "Uric acid: neuroprotective or neurotoxic?". Stroke 39 (5): e88; author reply e89. doi:10.1161/STROKEAHA.107.513242. PMID 18369163.
- ↑ Hayden MR, Tyagi SC (2004). "Uric acid: A new look at an old risk marker for cardiovascular disease, metabolic syndrome, and type 2 diabetes mellitus: The urate redox shuttle". Nutrition & Metabolism 1 (1): 10. doi:10.1186/1743-7075-1-10. PMID 15507132.
- ↑ Cutler RG (December 1984). "Urate and ascorbate: their possible roles as antioxidants in determining longevity of mammalian species". Archives of Gerontology and Geriatrics 3 (4): 321–48. doi:10.1016/0167-4943(84)90033-5. PMID 6532339.
- ↑ Gout Causes: List of Diet/Food Sources High or Low in Purine Content
- ↑ Gout Diet / Low Purine Diet - Limit High Purine foods
- ↑ Choi HK, Atkinson K, Karlson EW, Willett W, Curhan G (March 2004). "Purine-rich foods, dairy and protein intake, and the risk of gout in men". The New England Journal of Medicine 350 (11): 1093–103. doi:10.1056/NEJMoa035700. PMID 15014182.
- ↑ Edelson, Ed (2009-10-13). Common Antioxidant Might Slow Parkinson's. HealthDay, 13 October 2009. Retrieved on 24 October 2009 from http://news.yahoo.com/s/hsn/20091021/hl_hsn/commonantioxidantmightslowparkinsons.
External links
- MedlinePlus - Uric Acid Test
- International Kidney Stone Institute
- Purine content in food
|
|||||||||||||||||||||||||||
|
|||||