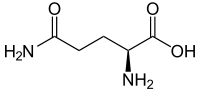
Glutamine
| L-Glutamine | |
|---|---|
 |
|
 |
|
|
Glutamine
|
|
|
Other names
2-Amino-4-carbamoylbutanoic acid
|
|
| Identifiers | |
| Abbreviations | Gln, Q |
| CAS number | 56-85-9 |
| PubChem | 738 |
| ChemSpider | 718 |
| EC-number | 200-292-1 |
| IUPHAR ligand | 723 |
|
SMILES
N[C@@H](CCC(N)=O)C(O)=O
|
|
| Properties[1] | |
| Molecular formula | C5H10N2O3 |
| Molar mass | 146.14 g mol−1 |
| Melting point |
185–186 ºC decomp. |
| Solubility in water | soluble |
| Chiral rotation [α]D | +6.5º (H2O, c = 2) |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) |
|
| Infobox references | |
Glutamine (abbreviated as Gln or Q) is one of the 20 amino acids encoded by the standard genetic code. Its side-chain is an amide formed by replacing the side-chain hydroxyl of glutamic acid with an amine functional group. Therefore, it can be considered the amide of glutamic acid. Its codons are CAA and CAG. In human blood, glutamine is the most abundant free amino acid, with a concentration of about 500-900 µmol/l.[2]
Contents |
Betain structure

Functions
Glutamine plays a role in a variety of biochemical functions including:
- Protein synthesis, as any other amino acid.
- Regulation of acid-base balance in the kidney by producing ammonium.[3]
- Cellular energy, as a source, next to glucose.[4]
- Nitrogen donation for many anabolic processes.[2]
- Carbon donation, as a source, refilling the citric acid cycle.[5]
Producing and consuming organs
Producers
Glutamine is synthesized by the enzyme glutamine synthetase from glutamate and ammonia. The most relevant glutamine-producing tissue is the muscle mass, accounting for about 90% of all glutamine synthesized. Glutamine is also released, in small amounts, by the lung and the brain.[6] Although the liver is capable of relevant glutamine synthesis, its role in glutamine metabolism is more regulatory than producing, since the liver takes up large amounts of glutamine derived from the gut.[2]
Consumers
The most eager consumers of glutamine are the cells of intestines[2], the kidney cells for the acid base balance, activated immune cells[7] and many cancer cells.[5] In respect to the last point mentioned, different glutamine analogues such as DON, Azaserine or Acivicin are tested as anti-cancer drugs.
Examples for the usage of glutamine
In catabolic states of injury and illness, glutamine becomes conditionally-essential (requiring intake from food or supplements). Glutamine has been studied extensively over the past 10–15 years and has been shown to be useful in treatment of serious illnesses, injury, trauma, burns, and treatment-related side-effects of cancer as well as in wound healing for postoperative patients.[8] Glutamine is also marketed as a supplement used for muscle growth in weightlifting, bodybuilding, endurance, and other sports, Evidence indicates that glutamine when orally loaded may increase plasma hGH levels by stimulating the anterior pitutitary gland. [9]. In biological research, L-glutamine is commonly added [10]to the media in cell culture.
Aiding recovery after surgery
It is also known that glutamine has various effects in reducing healing time after operations. Hospital-stay times after abdominal surgery can be reduced by providing parenteral nutrition regimes containing high amounts of glutamine to patients. Clinical trials have revealed that patients on supplementation regimes containing glutamine have improved nitrogen balances, generation of cysteinyl-leukotrienes from polymorphonuclear neutrophil granulocytes, and improved lymphocyte recovery and intestinal permeability (in postoperative patients), in comparison to those that had no glutamine within their dietary regime, all without any side-effects.[11]
Nutrition
Occurrences in nature
Glutamine is the most abundant naturally occurring, non-essential amino acid in the human body and one of the few amino acids that directly cross the blood-brain barrier.[12] In the body, it is found circulating in the blood as well as stored in the skeletal muscles. It becomes conditionally essential (requiring intake from food or supplements) in states of illness or injury.[8]
Dietary sources
Dietary sources of L-glutamine include beef, chicken, fish, eggs, milk, dairy products, wheat, cabbage, beets, beans, spinach, and parsley. Small amounts of free L-glutamine are also found in vegetable juices and fermented foods, such as miso.[13]
Aiding gastrointestinal function
In recent studies, glutamine-enriched diets have been linked with intestinal effects including maintenance of gut barrier function and cell differentiation. This may relate to the fact that the intestinal extraction rate of glutamine is higher than that for other amino acids, and is therefore thought to be the most viable option when attempting to alleviate conditions relating to the gastrointestinal tract. These conditions were discovered after comparing plasma concentration within the gut between glutamine-enriched and non-glutamine-enriched diets. However, even though glutamine is thought to have "cleansing" properties and effects, it is unknown to what extent glutamine has clinical benefits, due to the varied concentrations of glutamine in varieties of food.[14]
See also
- Isoglutamine
References
- ↑ Weast, Robert C., ed. (1981), CRC Handbook of Chemistry and Physics (62nd ed.), Boca Raton, FL: CRC Press, p. C-311, ISBN 0-8493-0462-8.
- ↑ 2.0 2.1 2.2 2.3 Brosnan, John T. (2003), "Interorgan amino acid transport and its regulation", J. Nutr. 133 (6): 2068S–72S, PMID 12771367, http://jn.nutrition.org/cgi/content/full/133/6/2068S.
- ↑ Guyton, Arthur C.; Hall, John E. (2005), Textbook of Medical Physiology (11th ed.), Saunders, p. 393, ISBN 0721602401.
- ↑ J. Carlos, Aledo (2004), "Glutamine breakdown in rapidly dividing cells: waste or investment?", BioEssays 26 (7): 778–85, doi:10.1002/bies.20063, PMID 15221859.
- ↑ 5.0 5.1 Yuneva, Mariia; Zamboni, Nicola; Oefner, Peter; Sachidanandam, Ravi; Lazebnik, Yuri (2007), "Defiency in glutamine but not glucose induces MYC-dependent apoptosis in human cells", J. Cell Biol. 178 (1): 93–105, doi:10.1083/jcb.200703099, PMID 17606868, PMC 2064426, http://jcb.rupress.org/cgi/content/full/178/1/93.
- ↑ Newsholme, P.; Lima, M. M. R.; Procopio, J.; Pithon-Curi, T. C.; Doi, S. Q.; Bazotte, R. B.; Curi, R. (2003), "Glutamine and glutamate as vital metabolites", Braz. J. Med. Biol. Res. 36 (2): 153–63, doi:10.1590/S0100-879X2003000200002, PMID 12563517, http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-879X2003000200002&lng=en&nrm=iso&tlng=en.
- ↑ Newsholme, Philip (2001), "Why Is L-Glutamine Metabolism Important to Cells of the Immune System in Health, Postinjury, Surgery or Infection?", J. Nutr. 131 (9): 2515S–22S, PMID 11533304, http://jn.nutrition.org/cgi/content/full/131/9/2515S.
- ↑ 8.0 8.1 Glutamine, University of Maryland Medical Center, http://www.umm.edu/altmed/articles/glutamine-000307.htm, retrieved 2009-09-06.
- ↑ {{citation |first1 = TC | last1 = Welbourne | title = Increased plasma bicarbonate and growth hormone after an oral glutamine load | location = American Journal of Clinical Nutrition, Vol 61, 1058-1061, Copyright © 1995 by The American Society for Clinical Nutrition, Inc | publisher = American Journal of Clinical Nutrition | year = 1995 | url=http://www.ajcn.org/cgi/content/abstract/61/5/1058.
- ↑ "L-glutamine", exactantigen.com, http://www.exactantigen.com/review/L-glutamine.html, retrieved 2009-11-27
- ↑ Morlion, Bart J.; Stehle, Peter; Wachtler, Paul; Siedhoff, Hans-P.; Köller, Manfred; König, Wolfgang; Fürst, Peter; Puchstein, Christoph (1998), "Total parenteral nutrition with glutamine dipeptide after major abdominal surgery: a randomized, double-blind, controlled study", Ann. Surg. 227 (2): 302–8, doi:10.1097/00000658-199802000-00022, PMID 9488531, PMC 1191250, http://www.annalsofsurgery.com/pt/re/annos/fulltext.00000658-199802000-00022.htm.
- ↑ Lee, Wha-Joon; Hawkins, Richard A.; Viña, Juan R.; Peterson, Darryl R. (1998), "Glutamine transport by the blood-brain barrier: a possible mechanism for nitrogen removal", Am. J. Physiol. Cell Physiol. 274 (4): C1101–7, PMID 9580550, http://ajpcell.physiology.org/cgi/content/full/274/4/C1101.
- ↑ "Glutamine", Vitamins Supplements Guide, http://www.vitamins-supplements.org/amino-acids/glutamine.php, retrieved 2007-11-01.
- ↑ Jian, Zhu Ming; Cao, J. D.; Zhu, X. G.; Zhao, W. X.; Yu, J. C.; Ma, E. L.; Wang, X. R.; Zhu, M. W. et al. (1999), "The impact of alanyl-glutamine on clinical safety, nitrogen balance, intestinal permeability, and clinical outcome in postoperative patients: a randomized, double-blind, controlled study of 120 patients", J. Parenteral Enteral Nutr. 23 (5 Suppl): S62–66, doi:10.1177/014860719902300516, PMID 10483898.
External links
|
||||||||||||||||||||||||||||||||||
|
||||||||||||||
|
||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||