Ester
Esters are chemical compounds derived by reacting an oxoacid (one containing an oxo group, X=O) with a hydroxyl compound such as an alcohol or phenol.[1] Esters are usually derived from an inorganic acid or organic acid in which at least one -OH (hydroxyl) group is replaced by an -O-alkyl (alkoxy) group, and most commonly from carboxylic acids and alcohols. Basically, esters are formed by condensing an acid with an alcohol.
Esters are ubiquitous. Many naturally occurring fats and oils are the fatty acid esters of glycerol. Esters with low molecular weight are commonly used as fragrances and found in essential oils and pheromones. Phosphoesters form the backbone of DNA molecules. Nitrate esters, such as nitroglycerin, are known for their explosive properties, while polyesters are important plastics, with monomers linked by ester moieties.
Contents |
Nomenclature
The names for esters are derived from the names of the parent alcohol and the carboxylic acid. For esters derived from the simplest carboxylic acids, the traditional names are used, such as formate, acetate, propionate, and butyrate. For esters from more complex carboxylic acids, the systematic name for the acid is used, followed by the suffix -oate. For example, hexyl octanoate, also called hexyl caprylate, has the formula CH3(CH2)6CO2(CH2)5CH3.
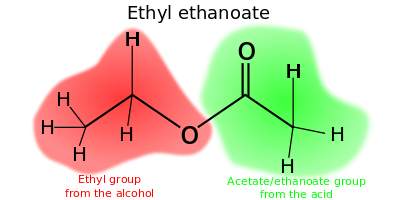
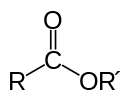
The chemical formulas of esters are typically written in the format of RCO2R', where R and R' are the organic parts of the carboxylic acid and alcohol respectively. For example butyl acetate, derived from butanol and acetic acid would be written CH3CO2C4H9. Alternative presentations are common including BuOAc and CH3COOC4H9.
Cyclic esters are called lactones. One example of many is valerolactone.
Orthoesters
An uncommon class of organic esters are the orthoesters, which have the formula RC(OR')3. Triethylorthoformate (HC(OC2H5)3) is derived, in terms of its name (but not its synthesis) from orthoformic acid (HC(OH)3) and ethanol.
"Inorganic esters"
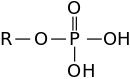
Ester is a general term for the product derived from the condensation of an oxo acid and an alcohol. Thus, the nomenclature extends to inorganic acids, especially phosphoric acid, sulfuric acid, and nitric acid. Cyclic esters are called lactones. The preparation of an ester is known generally as an esterification reaction. For example, triphenyl phosphate is the ester derived from phosphoric acid and phenol. Organic carbonates, such as ethylene carbonate are derived from carbonic acid and ethylene glycol. The term boronic ester is widely used but these compounds are not formally esters because they lack a B=O bond.
Structure and bonding
Esters contain a carbonyl center, which gives rise to 120° C-C-O and O-C-O angles. Unlike amides, esters are structurally flexible functional groups because rotation about the C-O-C bonds has a low barrier. Their flexibility and low polarity is manifested in their physical properties; they tend to be less rigid (lower melting point) and more volatile (lower boiling point) than the corresponding amides.
Physical properties and characterization
Esters are more polar than ethers but less polar than alcohols. They participate in hydrogen bonds as hydrogen-bond acceptors, but cannot act as hydrogen-bond donors, unlike their parent alcohols. This ability to participate in hydrogen bonding confers some water-solubility. Because of their lack of hydrogen-bond-donating ability, esters do not self-associate. Consequently esters are more volatile than carboxylic acids of similar molecular weight.
Characterization and analysis
Esters are usually identified by gas chromatography, taking advantage of their volatility. IR spectra for esters feature an intense sharp band in the range 1730-1750 cm-1 assigned to νC=O. This peak changes depending on the functional groups attached to the carbonyl. For example, a benzene ring or double bond in conjugation with the carbonyl will bring the wavenumber down about 30 cm-1.
Applications and occurrence
Esters are widespread in nature and are widely used in industry. In nature, fats are generally triesters derived from glycerol and fatty acids. Esters are responsible for the aroma of many fruits, including apples, pears, bananas, pineapples, and strawberries.[2] Several billion kilograms of polyesters are produced industrially annually, important products being polyethylene terephthalate, acrylate esters, and cellulose acetate.[3]
Preparation
Fischer esterification
The classic synthesis is the Fischer esterification, which involves treating a carboxylic acid with an alcohol in the presence of a dehydrating agent:
- R1COOH + R2OH
 R1COOR2 + H2O
R1COOR2 + H2O
Strong acids, typically sulfuric acid, catalyze this reaction. Many other acids are also used. Esterification is highly reversible. The yield of the product may be improved using le Chatelier's principle:
- using the alcohol in large excess (i.e. as a solvent),
- using a dehydrating agent. Sulfuric acid not only catalyzes the reaction but sequesters water (a reaction product). Other drying agents like molecular sieves can also be used.
- removal of water by physical means such as distillation as a low-boiling azeotropes with toluene, in conjunction with a Dean-Stark apparatus.
Alcoholysis of acyl chlorides and acid anhydrides
Alcohols react with acyl chlorides or acid anhydrides to give esters:
- RCOCl + R'OH → RCOOR' + HCl
- (RCO)2O + R'OH → RCO2R' + RCO2H
These reactions are irreversible, simplifying work-up. Since, acyl chlorides and acid anhydrides react also with water, anhydrous conditions are preferred. The analogous acylations of amines to give amides are less sensitive because amines are stronger nucleophiles and react more rapidly.
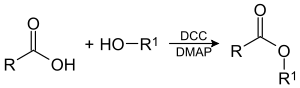
Steglich esterification
The Steglich esterification is method of forming esters under mild conditions. The method is especially popular in peptide synthesis, where the substrates are sensitive to harsh conditions like high heat. DCC (dicyclohexylcarbodiimide) is used to activate the carboxylic acid to further reaction. DMAP (4-dimethylaminopyridine) is used catalytically as an acyl-transfer agent.
Other methods
- Transesterifications between other esters. This method is widely used industrially.
- Favorskii rearrangement of α-haloketones in presence of base
- Nucleophilic displacement of alkyl halides with carboxylic acid salts.
- Baeyer-Villiger oxidation of ketones with peroxides.
- Pinner reaction of nitriles with an alcohol.
Reactions
Esters react primarily at one of two locations, the carbonyl at the carbon adjacent the carbonyl group. The carbonyl is weakly electrophilic and is attacked by strong nucleophilies (amines, alkoxides, hydride sources, organolithium compounds, etc). The C-H bonds adjacent to the carbonyl are weakly acidic but undergo deprotonation with strong bases. This process that usually initiates condensation reactions.
Nucleophilic substitutions
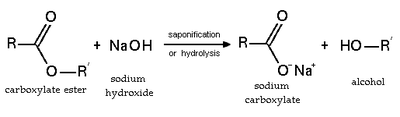
Esters undergo hydrolysis under acid and basic conditions. Under acid conditions, the reaction is the reverse reaction of the Fischer esterification. Under basic conditions, hydroxide acts as a nucleophile, while an alkoxide is the leaving group. This reaction, saponification, is the basis of soap making.
The alkoxide group may also be displaced by stronger nucleophiles such as ammonia or primary or secondary amines to give amides.
Reduction
Esters are relatively resistant to reduction. Lithium aluminium hydride is able to reduce esters to two primary alcohols, whereas sodium borohydride does so more slowly. DIBAH reduces esters to aldehydes, while forcing conditions are required for hydrogenation.[4] In the Bouveault-Blanc reduction, sodium in the presence of proton sources is the reducing agent. Such reactions can also be conducted by hydrogenation.
-
- RCO2R' + 2 H2 → RCH2OH + R'OH
Reactions adjacent the carboxyl group
Similar to carbonyl compounds, the hydrogen atoms on the carbon adjacent ("α to") the carboxyl group are acidic. They may be removed by relatively strong bases, such as an alkoxide salt. Deprotonating the alpha position gives a nucleophile, which may further react, e.g. the Claisen condensation and its intramolecular equivalent, the Dieckmann condensation. This property is exploited in the malonic ester synthesis, where the diester of malonic acid reacts with an electrophile (e.g. alkyl halide), and is subsequently decarboxylated. This is a two carbon homologation reaction.
Other reactions
- Phenyl esters react to hydroxyarylketones in the Fries rearrangement.
- Specific esters are functionalized with an α-hydroxyl group in the Chan rearrangement.
- Esters are converted to isocyanates through intermediate hydroxamic acids in the Lossen rearrangement.
- Esters with β-hydrogen atoms can be converted to alkenes in ester pyrolysis.
Protecting groups
As a class, esters serve as protecting groups for carboxylic acids. Protecting a carboxylic acid is useful in peptide synthesis, to prevent self-reactions of the bifunctional amino acids. Methyl and ethyl esters are commonly available for many amino acids; the t-butyl ester tends to be more expensive. However, t-butyl esters are particularly useful because under strongly acidic conditions, the t-butyl esters undergo elimination to give the carboxylic acid and isobutene, simplifying work-up.
External links
- An introduction to esters
- Molecule of the month: Ethyl acetate and other esters
- An example of esters commercial application
References
- ↑ IUPAC Gold Book internet edition: "esters".
- ↑ McGee, Harold. On Food and Cooking. 2003, Scribner, New York.
- ↑ Wilhelm Riemenschneider1 and Hermann M. Bolt "Esters, Organic" Ullmann's Encyclopedia of Industrial Chemistry, 2005, Wiley-VCH, Weinheim. doi:10.1002/14356007.a09_565.pub2
- ↑ W. Reusch. "Carboxyl Derivative Reactivity". Virtual Textbook of Organic Chemistry. http://www.cem.msu.edu/~reusch/VirtualText/crbacid2.htm#react2.
Appendix of ester odorants
Many esters have distinctive fruit-like odors, which has led to their commonplace use in artificial flavorings and fragrances.
| Ester Name | Structure | Odor or occurrence |
|---|---|---|
| Allyl hexanoate | pineapple | |
| Benzyl acetate | pear, strawberry, jasmine | |
| Bornyl acetate |  |
pine tree flavor |
| Butyl butyrate | pineapple | |
| Ethyl acetate | nail polish remover, model paint, model airplane glue | |
| Ethyl butyrate | banana, pineapple, strawberry | |
| Ethyl hexanoate | pineapple, waxy-green banana | |
| Ethyl cinnamate | cinnamon | |
| Ethyl formate | lemon, rum, strawberry | |
| Ethyl heptanoate | apricot, cherry, grape, raspberry | |
| Ethyl isovalerate | apple | |
| Ethyl lactate | 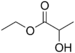 |
butter, cream |
| Ethyl nonanoate | grape | |
| Ethyl pentanoate | apple | |
| Geranyl acetate | geranium | |
| Geranyl butyrate | cherry | |
| Geranyl pentanoate | apple | |
| Isobutyl acetate | cherry, raspberry, strawberry | |
| Isobutyl formate | raspberry | |
| Isoamyl acetate | pear, banana (flavoring in Pear drops) | |
| Isopropyl acetate | fruity | |
| Linalyl acetate | lavender, sage | |
| Linalyl butyrate | peach | |
| Linalyl formate | apple, peach | |
| Methyl acetate | 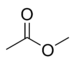 |
glue |
| Methyl anthranilate | 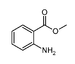 |
grape, jasmine |
| Methyl benzoate | fruity, ylang ylang, feijoa | |
| Methyl butyrate (methyl butanoate) | pineapple, apple, strawberry | |
| Methyl cinnamate | strawberry | |
| Methyl pentanoate (methyl valerate) | flowery | |
| Methyl phenylacetate | honey | |
| Methyl salicylate (oil of wintergreen) | 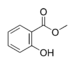 |
Modern root beer, wintergreen, Germolene and Ralgex ointments (UK) |
| Nonyl caprylate | orange | |
| Octyl acetate | fruity-orange | |
| Octyl butyrate | parsnip | |
| Amyl acetate (pentyl acetate) | apple, banana | |
| Pentyl butyrate (amyl butyrate) | apricot, pear, pineapple | |
| Pentyl hexanoate (amyl caproate) | apple, pineapple | |
| Pentyl pentanoate (amyl valerate) | apple | |
| Propyl ethanoate | pear | |
| Propyl isobutyrate | rum | |
| Terpenyl butyrate | 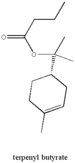 |
cherry |
|
||||||||