Circadian rhythm
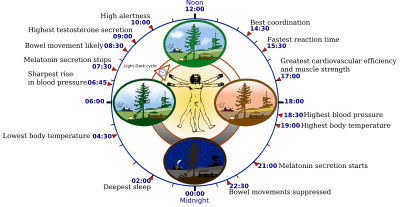
A circadian rhythm is a roughly 24-hour cycle in the biochemical, physiological, or behavioural processes of living entities, including plants, animals, fungi and cyanobacteria (see bacterial circadian rhythms). The term "circadian" comes from the Latin circa, "around", and diem or dies, "day", meaning literally "approximately one day". The formal study of biological temporal rhythms such as daily, tidal, weekly, seasonal, and annual rhythms, is called chronobiology.
Although circadian rhythms are endogenous, they are adjusted (entrained) to the environment by external cues called zeitgebers, the primary one of which is daylight.
Contents |
History
The earliest known account of a circadian process dates from the 4th century BC, when Androsthenes, a ship captain serving under Alexander the Great, described diurnal leaf movements of the tamarind tree.[1] The first modern observation of endogenous circadian oscillation was by the French scientist Jean-Jacques d'Ortous de Mairan in the 1700s; he noted that 24-hour patterns in the movement of the leaves of the plant Mimosa pudica continued even when the plants were kept in constant darkness.[2]
In 1918, J.S. Szymanski showed that animals are capable of maintaining 24-hour activity patterns in the absence of external cues such as light and changes in temperature.[3] Joseph Takahashi discovered the genetic basis for the rodent circadian rhythm in 1994.[4][5]
The term "circadian" was coined by Franz Halberg in the late 1950s.[6]
Criteria
Historically, to differentiate genuinely endogenous circadian rhythms from coincidental or apparent ones, three general criteria must be met: 1) the rhythms persist in the absence of cues, 2) they persist equally precisely over a range of temperatures, and 3) the rhythms can be adjusted to match the local time:
- The rhythm persists in constant conditions (for example, constant dark) with a period of about 24 hours. The rationale for this criterion is to distinguish circadian rhythms from those "apparent" rhythms that are merely responses to external periodic cues. A rhythm cannot be declared to be endogenous unless it has been tested in conditions without external periodic input.
- The rhythm is temperature-compensated, i.e., it maintains the same period over a range of temperatures. The rationale for this criterion is to distinguish circadian rhythms from other biological rhythms arising due to the circular nature of a reaction pathway. At a low enough or high enough temperature, the period of a circular reaction may reach 24 hours, but it will be merely coincidental.
- The rhythm can be reset by exposure to an external stimulus. The rationale for this criterion is to distinguish circadian rhythms from other imaginable endogenous 24-hour rhythms that are immune to resetting by external cues and, hence, do not serve the purpose of estimating the local time. Travel across time zones illustrates the necessity of the ability to adjust the biological clock so that it can reflect the local time and anticipate what will happen next. Until rhythms are reset, a person usually experiences jet lag.
However, criterion 2 (that circadian rhythms persist equally precisely over a range of temperatures) is now understood to not be a factor. Thermal energy will affect the kinetics of all molecular processes, and likewise, temperature changes applied to the circadian clock mechanisms of many organisms (including fungi and even the SCN of mammals) has been shown to affect the frequency of the rhythm, as would be expected. In some instances, heat can provide a stronger zeitgeber than light.
Origin
Photosensitive proteins and circadian rhythms are believed to have originated in the earliest cells, with the purpose of protecting the replicating of DNA from high ultraviolet radiation during the daytime. As a result, replication was relegated to the dark. The fungus Neurospora, which exists today, retains this clock-regulated mechanism.
Circadian rhythms allow organisms to anticipate and prepare for precise and regular environmental changes; they have great value in relation to the outside world. The rhythmicity appears to be as important in regulating and coordinating internal metabolic processes, as in coordinating with the environment.[7] This is suggested by the maintenance (heritability) of circadian rhythms in fruit flies after several hundred generations in constant laboratory conditions,[8] as well as in creatures in constant darkness in the wild, and by the experimental elimination of behavioural but not physiological circadian rhythms in quail.[9][10]
The simplest known circadian clock is that of the prokaryotic cyanobacteria. Recent research has demonstrated that the circadian clock of Synechococcus elongatus can be reconstituted in vitro with just the three proteins of their central oscillator. This clock has been shown to sustain a 22-hour rhythm over several days upon the addition of ATP. Previous explanations of the prokaryotic circadian timekeeper were dependent upon a DNA transcription/translation feedback mechanism.
In 1971, Ronald J. Konopka and Seymour Benzer first identified a genetic component of the biological clock using the fruit fly as a model system. Three mutant lines of flies displayed aberrant behaviour: one had a shorter period, another had a longer one, and the third had none. All three mutations mapped to the same gene, which was named "period".[11] The same gene was identified to be defective in the sleep disorder FASPS (Familial advanced sleep phase syndrome) in human beings thirty years later, underscoring the conserved nature of the molecular circadian clock through evolution. Many more genetic components of the biological clock are now known. Their interactions result in an interlocked feedback loop of gene products resulting in periodic fluctuations that the cells of the body interpret as a specific time of the day.
A great deal of research on biological clocks was done in the latter half of the 20th century. It is now known that the molecular circadian clock can function within a single cell; i.e., it is cell-autonomous.[12] At the same time, different cells may communicate with each other resulting in a synchronised output of electrical signaling. These may interface with endocrine glands of the brain to result in periodic release of hormones. The receptors for these hormones may be located far across the body and synchronise the peripheral clocks of various organs. Thus, the information of the time of the day as relayed by the eyes travels to the clock in the brain, and, through that, clocks in the rest of the body may be synchronised. This is how the timing of, for example, sleep/wake, body temperature, thirst, and appetite are coordinately controlled by the biological clock.
Importance in animals
Circadian rhythmicity is present in the sleeping and feeding patterns of animals, including human beings. There are also clear patterns of core body temperature, brain wave activity, hormone production, cell regeneration and other biological activities. In addition, photoperiodism, the physiological reaction of organisms to the length of day or night, is vital to both plants and animals, and the circadian system plays a role in the measurement and interpretation of day length.
| “ | Timely prediction of seasonal periods of weather conditions, food availability or predator activity is crucial for survival of many species. Although not the only parameter, the changing length of the photoperiod ('daylength') is the most predictive environmental cue for the seasonal timing of physiology and behavior, most notably for timing of migration, hibernation and reproduction.[13] | ” |
Impact of light–dark cycle
The rhythm is linked to the light–dark cycle. Animals, including humans, kept in total darkness for extended periods eventually function with a freerunning rhythm. Each "day", their sleep cycle is pushed back or forward, depending on whether their endogenous period is shorter or longer than 24 hours. The environmental cues that each day reset the rhythms are called zeitgebers (from the German, time-givers).[14] It is interesting to note that totally-blind subterranean mammals (e.g., blind mole rat Spalax sp.) are able to maintain their endogenous clocks in the apparent absence of external stimuli. Although they lack image-forming eyes, their photoreceptors (detect light) are still functional; as well, they do surface periodically.[15]
Freerunning organisms that normally have one or two consolidated sleep episodes will still have them when in an environment shielded from external cues, but the rhythm is, of course, not entrained to the 24-hour light/dark cycle in nature. The sleep–wake rhythm may, in these circumstances, become out of phase with other circadian or ultradian rhythms such as metabolic, hormonal, CNS electrical, or neurotransmitter rhythms.[16]
Recent research has influenced the design of spacecraft environments, as systems that mimic the light/dark cycle have been found to be highly beneficial to astronauts.
Arctic animals
Norwegian researchers at the University of Tromsø have shown that some Arctic animals (ptarmigan, reindeer) show circadian rhythms only in the parts of the year that have daily sunrises and sunsets. In one study of reindeer, animals at 70 degrees North showed circadian rhythms in the autumn, winter, and spring, but not in the summer. Reindeer at 78 degrees North showed such rhythms only autumn and spring. The researchers suspect that other Arctic animals as well may not show circadian rhythms in the constant light of summer and the constant dark of winter.[17][18]
However, another study in northern Alaska found that ground squirrels and porcupines strictly maintained their circadian rhythms through 82 days and nights of sunshine. The researchers speculate that these two small mammals see that the apparent distance between the sun and the horizon is shortest once a day, and, thus, a sufficient signal to adjust by.[19]
Butterfly migration
The navigation of the fall migration of the Eastern North American monarch butterfly (Danaus plexippus) to their overwintering grounds in central Mexico uses a time-compensated sun compass that depends upon a circadian clock in their antennae.[20][21]
Biological clock in mammals

The primary circadian "clock" in mammals is located in the suprachiasmatic nucleus (or nuclei) (SCN), a pair of distinct groups of cells located in the hypothalamus. Destruction of the SCN results in the complete absence of a regular sleep–wake rhythm. The SCN receives information about illumination through the eyes. The retina of the eye contains not only the "classical" photoreceptors which are used for vision but ganglion cells which respond to light and are called photosensitive ganglion cells.
These cells contain the photo pigment melanopsin and their signals follow a pathway called the retinohypothalamic tract, leading to the SCN. If cells from the SCN are removed and cultured, they maintain their own rhythm in the absence of external cues.
The SCN takes the information on the lengths of the day and night from the retina, interprets it, and passes it on to the pineal gland, a tiny structure shaped like a pine cone and located on the epithalamus. In response, the pineal secretes the hormone melatonin. Secretion of melatonin peaks at night and ebbs during the day and its presence provides information about night-length.
The circadian rhythms of humans can be entrained to slightly shorter and longer periods than the Earth's 24 hours. Researchers at Harvard have recently shown that human subjects can at least be entrained to a 23.5-hour cycle and a 24.65-hour cycle (the latter being the natural solar day-night cycle on the planet Mars).[22]
Determining the human circadian rhythm
The classic phase markers for measuring the timing of a mammal's circadian rhythm are:[23]
- melatonin secretion by the pineal gland
- core body temperature.
For temperature studies, people must remain awake but calm and semi-reclined in near darkness while their rectal temperatures are taken continuously. The average human adult's temperature reaches its minimum at about 05:00 (5 a.m.), about two hours before habitual wake time, though variation is great among normal chronotypes.
Melatonin is absent from the system or undetectably low during daytime. Its onset in dim light, dim-light melatonin onset (DLMO), at about 21:00 (9 p.m.) can be measured in the blood or the saliva. Its major metabolite can also be measured in morning urine. Both DLMO and the midpoint (in time) of the presence of the hormone in the blood or saliva have been used as circadian markers.
However, newer research indicates that the melatonin offset may be the most reliable marker. Benloucif et al. in Chicago in 2005 found that melatonin phase markers were more stable and more highly correlated with the timing of sleep than the core temperature minimum. They found that both sleep offset and melatonin offset were more strongly correlated with the various phase markers than sleep onset. In addition, the declining phase of the melatonin levels was more reliable and stable than the termination of melatonin synthesis.[23]
One method used for measuring melatonin offset is to analyse a sequence of urine samples throughout the morning for the presence of the melatonin metabolite 6-sulphatoxymelatonin (aMT6s). Laberge et al. in Quebec in 1997 used this method in a study that confirmed the frequently found delayed circadian phase in healthy adolescents.[24]
Outside the "master clock"
More-or-less independent circadian rhythms are found in many organs and cells in the body outside the suprachiasmatic nuclei (SCN), the "master clock". These clocks, called peripheral oscillators, are found in the oesophagus, lungs, liver, pancreas, spleen, thymus, and the skin.[25] Though oscillators in the skin respond to light, a systemic influence has not been proven so far.[26][27] There is also some evidence that the olfactory bulb and prostate may experience oscillations when cultured, suggesting that these structures may also be weak oscillators.
Furthermore, liver cells, for example, appear to respond to feeding rather than to light. Cells from many parts of the body appear to have freerunning rhythms.
Light and the biological clock
Light resets the biological clock in accordance with the phase response curve (PRC). Depending on the timing, light can advance or delay the circadian rhythm. Both the PRC and the required illuminance vary from species to species and lower light levels are required to reset the clocks in nocturnal rodents than in humans.
Lighting levels that affect the circadian rhythm in humans are higher than the levels usually used in artificial lighting in homes. According to some researchers[28] the illumination intensity that excites the circadian system has to reach up to 1000 lux striking the retina. In addition to light intensity, wavelength (or colour) of light is a factor in the entrainment of the body clock. Melanopsin is most efficiently excited by blue light, 420–440 nm[29] according to some researchers while others have reported 470–485 nm.
It is thought that the direction of the light may have an effect on entraining the circadian rhythm;[28] light coming from above, resembling an image of a bright sky, has greater effect than light entering our eyes from below.
According to a 2010 study completed by the Lighting Research Center, daylight has a direct effect on circadian rhythms and, consequently, on performance and well-being. The research showed that students who experience disruption in lighting schemes in the morning consequently experience disruption in sleeping patterns. The change in sleeping patterns may lead to negatively impacted student performance and alertness. Removing circadian light in the morning delays the dim light melatonin onset by 6 minutes a day, for a total of 30 minutes for five days.[30]
Enforced longer cycles
Modern research under very controlled conditions has shown the human period for adults to be just slightly longer than 24 hours on average. Czeisler et al. at Harvard found the range for normal, healthy adults of all ages to be quite narrow: 24 hours and 11 minutes ± 16 minutes. The "clock" resets itself daily to the 24-hour cycle of the Earth's rotation.[31]
The 28-hour day is presented as a concept of time management.[32] It builds on the fact that the week of seven days at 24 hours and a "week" of six days at 28 hours both equal a week of 168 hours. To live on the 28-hour day and six-day week would require staying awake for 19 to 20 hours and sleeping for eight to nine hours. Each "day" on this system has a unique light/dark pattern.
Studies by Nathaniel Kleitman[33] in 1938 and by Derk-Jan Dijk and Charles Czeisler[34][35] in 1994/5 have put human subjects on enforced 28-hour sleep–wake cycles, in constant dim light and with other time cues suppressed, for over a month. Because normal people cannot entrain to a 28-hour day,[36] this is referred to as a forced desynchrony protocol. Sleep and wake episodes are uncoupled from the endogenous circadian period of about 24.18 hours and researchers are allowed to assess the effects of circadian phase on aspects of sleep and wakefulness including sleep latency and other functions.[37]
Early research into circadian rhythms suggested that most people preferred a day closer to 25 hours when isolated from external stimuli like daylight and timekeeping. Early investigators determined the human circadian period to be 25 hours or more. They went to great lengths to shield subjects from time cues and daylight, but they were not aware of the effects of indoor electric lights. The subjects were allowed to turn on light when they were awake and to turn it off when they wanted to sleep. Electric light in the evening delayed their circadian phase. These results became well known.[31] Researchers allowed subjects to keep electric lighting on in the evening, as it was thought at that time that a couple of 60W bulbs would not have a resetting effect on the circadian rhythms of humans. More recent research has shown that adults have a built-in day, which averages just over 24 hours, that indoor lighting does affect circadian rhythms and that most people attain their best-quality sleep during their chronotype-determined sleep periods.
Human health
Timing of medical treatment in coordination with the body clock may significantly increase efficacy and reduce drug toxicity or adverse reactions. For example, appropriately timed treatment with angiotensin converting enzyme inhibitors (ACEi) may reduce nocturnal blood pressure and also benefit left ventricular (reverse) remodelling.

A number of studies have concluded that a short period of sleep during the day, a power-nap, does not have any effect on normal circadian rhythm, but can decrease stress and improve productivity.[38][39]
There are many health problems associated with disturbances of the human circadian rhythm, such as seasonal affective disorder (SAD), delayed sleep phase syndrome (DSPS) and other circadian rhythm disorders.[40] Circadian rhythms also play a part in the reticular activating system, which is crucial for maintaining a state of consciousness. In addition, a reversal in the sleep–wake cycle may be a sign or complication of uremia,[41] azotemia or acute renal failure.
Studies have also shown that light has a direct effect on human health because of the way it influences the circadian rhythms.[42][43][44]
Circadian rhythm and airline pilots
Due to the work nature of airline pilots, who often traverse multiple timezones and regions of sunlight and darkness in one day, and spend many hours awake both day and night, they are often unable to maintain sleep patterns that correspond to the natural human circadian rhythm; this situation can easily lead to fatigue. The NTSB cites this situation as a contributing factor to many accidents[45] and has conducted multiple research studies in order to find methods of combating fatigue in pilots.[46][47]
Disruption
Disruption to rhythms usually has a negative effect. Many travellers have experienced the condition known as jet lag, with its associated symptoms of fatigue, disorientation and insomnia.
A number of other disorders, for example bipolar disorder and some sleep disorders, are associated with irregular or pathological functioning of circadian rhythms. Recent research suggests that circadian rhythm disturbances found in bipolar disorder are positively influenced by lithium's effect on clock genes.[48]
Disruption to rhythms in the longer term is believed to have significant adverse health consequences on peripheral organs outside the brain, particularly in the development or exacerbation of cardiovascular disease.[49] The suppression of melatonin production associated with the disruption of the circadian rhythm may increase the risk of developing cancer.[50]
Effect of drugs
Circadian rhythms and clock genes expressed in brain regions outside the SCN may significantly influence the effects produced by drugs such as cocaine.[51][52] Moreover, genetic manipulations of clock genes profoundly affect cocaine's actions.[53]
See also
- Actigraphy (also known as Actimetry)
- ARNTL
- ARNTL2
- Bacterial circadian rhythms
- Circadian rhythm sleep disorders
- Cryptochrome
- CRY1 and CRY2, the cryptochrome family genes
- Delayed sleep phase syndrome
- Diurnal cycle
- Jet lag
- Light effects on circadian rhythm
- Light in school buildings
- PER1, PER2, and PER3, the period family genes
References
Bibliography
- Aschoff J (ed.) (1965) Circadian Clocks. North Holland Press, Amsterdam
- Avivi A, Albrecht U, Oster H, Joel A, Beiles A, Nevo E (November 2001). "Biological clock in total darkness: the Clock/MOP3 circadian system of the blind subterranean mole rat". Proceedings of the National Academy of Sciences of the United States of America 98 (24): 13751–6. doi:10.1073/pnas.181484498. PMID 11707566.
- Avivi A, Oster H, Joel A, Beiles A, Albrecht U, Nevo E (September 2002). "Circadian genes in a blind subterranean mammal II: conservation and uniqueness of the three Period homologs in the blind subterranean mole rat, Spalax ehrenbergi superspecies". Proceedings of the National Academy of Sciences of the United States of America 99 (18): 11718–23. doi:10.1073/pnas.182423299. PMID 12193657.
- Ditty JL, Williams SB, Golden SS (2003). "A cyanobacterial circadian timing mechanism". Annual Review of Genetics 37: 513–43. doi:10.1146/annurev.genet.37.110801.142716. PMID 14616072.
- Dunlap JC, Loros J, DeCoursey PJ (2003) Chronobiology: Biological Timekeeping. Sinauer, Sunderland
- Dvornyk V, Vinogradova O, Nevo E (March 2003). "Origin and evolution of circadian clock genes in prokaryotes". Proceedings of the National Academy of Sciences of the United States of America 100 (5): 2495–500. doi:10.1073/pnas.0130099100. PMID 12604787.
- Koukkari WL, Sothern RB (2006) Introducing Biological Rhythms. Springer, New York
- Martino T, Arab S, Straume M, et al. (April 2004). "Day/night rhythms in gene expression of the normal murine heart". Journal of Molecular Medicine 82 (4): 256–64. doi:10.1007/s00109-003-0520-1. PMID 14985853.
- Refinetti R (2006) Circadian Physiology, 2nd ed. CRC Press, Boca Raton
- Takahashi JS, Zatz M (September 1982). "Regulation of circadian rhythmicity". Science 217 (4565): 1104–11. doi:10.1126/science.6287576. PMID 6287576.
- Tomita J, Nakajima M, Kondo T, Iwasaki H (January 2005). "No transcription-translation feedback in circadian rhythm of KaiC phosphorylation". Science 307 (5707): 251–4. doi:10.1126/science.1102540. PMID 15550625.
- Moore-Ede, Martin C.; Sulszman, Frank M.; Fuller, Charles A. (1982). The Clocks that Time Us: Physiology of the Circadian Timing System. Cambridge, MA: Harvard University Press. ISBN 0-674-13581-4.
Notes
- ↑ Bretzl H. (1903). Botanische Forschungen des Alexanderzuges. Leipzig: Teubner.
- ↑ Gardner MJ, Hubbard KE, Hotta CT, Dodd AN, Webb AA (July 2006). "How plants tell the time". The Biochemical Journal 397 (1): 15–24. doi:10.1042/BJ20060484. PMID 16761955. PMC 1479754. http://www.biochemj.org/bj/397/0015/bj3970015.htm. Retrieved 2010-06-11.
- ↑ Danchin, Antoine. "Important dates 1900-1919". HKU-Pasteur Research Centre (Paris). http://www.pasteur.fr/recherche/unites/REG/causeries/dates_1900.html. Retrieved 2008-01-12.
- ↑ "Gene Discovered in Mice that Regulates Biological Clock". Chicago Tribune. April 29, 1994.
- ↑ Vitaterna MH, King DP, Chang AM, et al. (April 1994). "Mutagenesis and mapping of a mouse gene, Clock, essential for circadian behavior". Science 264 (5159): 719–25. doi:10.1126/science.8171325. PMID 8171325.
- ↑ Zivkovic, Bora (May 3, 2008). "Circadian Quackery". A Blog Around the Clock. ScienceBlogs. http://scienceblogs.com/clock/2008/05/circadian_quackery_1.php. Retrieved 2010-02-23.
- ↑ Sharma VK (November 2003). "Adaptive significance of circadian clocks". Chronobiology International 20 (6): 901–19. doi:10.1081/CBI-120026099. PMID 14680135.
- ↑ Sheeba V, Sharma VK, Chandrashekaran MK, Joshi A (September 1999). "Persistence of eclosion rhythm in Drosophila melanogaster after 600 generations in an aperiodic environment". Die Naturwissenschaften 86 (9): 448–9. doi:10.1007/s001140050651. PMID 10501695.
- ↑ Guyomarc'h C, Lumineau S, Richard JP (May 1998). "Circadian rhythm of activity in Japanese quail in constant darkness: variability of clarity and possibility of selection". Chronobiology International 15 (3): 219–30. PMID 9653576.
- ↑ Zivkovic BD, Underwood H, Steele CT, Edmonds K (October 1999). "Formal properties of the circadian and photoperiodic systems of Japanese quail: phase response curve and effects of T-cycles". Journal of Biological Rhythms 14 (5): 378–90. PMID 10511005. http://jbr.sagepub.com/cgi/pmidlookup?view=long&pmid=10511005.
- ↑ Purves, Dale; Augustine, George J.; Fitzpatrick, David et al., eds (2001). "Molecular Mechanisms of Biological Clocks". Neuroscience. Sunderland, Mass: Sinauer Associates. pp. 666–7. ISBN 0-87893-742-0. http://www.ncbi.nlm.nih.gov/books/bv.fcgi?rid=neurosci.box.1963.
- ↑ Nagoshi E, Saini C, Bauer C, Laroche T, Naef F, Schibler U (November 2004). "Circadian gene expression in individual fibroblasts: cell-autonomous and self-sustained oscillators pass time to daughter cells". Cell 119 (5): 693–705. doi:10.1016/j.cell.2004.11.015. PMID 15550250.
- ↑ Zivkovic, Bora "Coturnix" (2005-08-13 / July 25, 2007). "Clock Tutorial #16: Photoperiodism - Models and Experimental Approaches". A Blog Around the Clock. ScienceBlogs. http://scienceblogs.com/clock/2007/07/clock_tutorial_16_photoperiodi_1.php. Retrieved 2007-12-09.
- ↑ Shneerson JM, Ohayon MM, Carskadon MA (2007). "Circadian rhythms". Rapid eye movement (REM) sleep. Armenian Medical Network. http://www.sleep.health.am/sleep/more/circadian-rhythms/. Retrieved 2007-09-19.
- ↑ "The Rhythms of Life: The Biological Clocks That Control the Daily Lives of Every Living Thing" Russell Foster & Leon Kreitzman, Publisher: Profile Books Ltd.
- ↑ Regestein, Quentin R.; Pavlova, Milena (September 1995). [http://www.sciencedirect.com/science/article/B6T70-3Y6PCPVF/ 2/d71146c55942bb86e95e87fe45e95687 "Treatment of delayed sleep phase syndrome"] (Abstract). General Hospital Psychiatry (Elsevier Science Inc.) 17 (5): 335-345. doi:10.1016/0163-8343(95)00062-V. http://www.sciencedirect.com/science/article/B6T70-3Y6PCPVF/ 2/d71146c55942bb86e95e87fe45e95687.
- ↑ Spilde, Ingrid (December 2005). "Reinsdyr uten døgnrytme" (in Language: Norwegian, Bokmål). forskning.no. http://www.forskning.no/Artikler/2005/desember/1135264557.29. Retrieved 2007-11-24.
- ↑ Zivkovic, Bora, aka Coturnix, chronobiologist. "Circadian Rhythms, or Not, in Arctic Reindeer". A Blog around the Clock. ScienceBlogs.com. http://scienceblogs.com/clock/2007/07/circadian_rhythms_or_not_in_ar_1.php. Retrieved 2007-11-24.
- ↑ Zivkovic, Bora, aka Coturnix, chronobiologist (2007-02-11). "Small Arctic Mammals Entrain to Something during the Long Summer Day". A Blog Around the Clock. ScienceBlogs.com. http://scienceblogs.com/clock/2007/02/small_arctic_mammals_entrain_t.php. Retrieved 2007-11-26.
- ↑ Merlin C, Gegear RJ, Reppert SM (September 2009). "Antennal circadian clocks coordinate sun compass orientation in migratory monarch butterflies". Science 325 (5948): 1700–4. doi:10.1126/science.1176221. PMID 19779201.
- ↑ Kyriacou CP (September 2009). "Physiology. Unraveling traveling". Science 325 (5948): 1629–30. doi:10.1126/science.1178935. PMID 19779177.
- ↑ Scheer FA, Wright KP, Kronauer RE, Czeisler CA (2007). "Plasticity of the intrinsic period of the human circadian timing system". PLos ONE 2 (1): e721. doi:10.1371/journal.pone.0000721. PMID 17684566.
- ↑ 23.0 23.1 Benloucif S, Guico MJ, Reid KJ, Wolfe LF, L'hermite-Balériaux M, Zee PC (April 2005). "Stability of melatonin and temperature as circadian phase markers and their relation to sleep times in humans". Journal of Biological Rhythms 20 (2): 178–88. doi:10.1177/0748730404273983. PMID 15834114.
- ↑ Laberge, L.; Lesperance, P.; Tremblay, R.; Lambert, C.; Montplaisir, J. (1997). "Phase delay of 6-sulphatoxymelatonin in normal adolescents". Sleep Research (Québec, Canada: Centre d'etude du Sommeil, Hopital du Sacre-Coeur, Département de Psychologie, Département de Pharmacologie, Departement de Psychiatrie, Université de Montréal) 26: 727. http://www.websciences.org/cftemplate/NAPS/archives/indiv.cfm?issn=19979287. Retrieved 2007-12-18.
- ↑ Zanello SB, Jackson DM, Holick MF (October 2000). "Expression of the circadian clock genes clock and period1 in human skin". The Journal of Investigative Dermatology 115 (4): 757–60. doi:10.1046/j.1523-1747.2000.00121.x. PMID 10998156.
- ↑ Kawara S, Mydlarski R, Mamelak AJ, et al. (December 2002). "Low-dose ultraviolet B rays alter the mRNA expression of the circadian clock genes in cultured human keratinocytes". The Journal of Investigative Dermatology 119 (6): 1220–3. doi:10.1046/j.1523-1747.2002.19619.x. PMID 12485420.
- ↑ Campbell SS, Murphy PJ (January 1998). "Extraocular circadian phototransduction in humans". Science 279 (5349): 396–9. doi:10.1126/science.279.5349.396. PMID 9430592.
- ↑ 28.0 28.1 Semjonova, Milena (2003). "Healthy Lighting, from a lighting designer's perspective". Milena Lighting Design. http://www.enlighter.org/images/2009/01/healthyLighting.pdf.
- ↑ Newman LA, Walker MT, Brown RL, Cronin TW, Robinson PR (November 2003). "Melanopsin forms a functional short-wavelength photopigment". Biochemistry 42 (44): 12734–8. doi:10.1021/bi035418z. PMID 14596587.
- ↑ Figueiro MG, Rea MS (February 2010). "Lack of short-wavelength light during the school day delays dim light melatonin onset (DLMO) in middle school students". Neuro Endocrinology Letters 31 (1): 4. PMID 20150866.
- ↑ 31.0 31.1 Charles A. Czeisler MD, PhD (1999). "Human Biological Clock Set Back an Hour". http://www.hno.harvard.edu/gazette/1999/07.15/bioclock24.html. Retrieved 2007-09-23. "The variation between our subjects, with a 95 percent level of confidence, was no more than plus or minus 16 minutes, a remarkably small range."
- ↑ Digital Beat Productions (1997). "28 Hour Day". http://www.dbeat.com/28/benefit2.htm. Retrieved 2008-02-19.
- ↑ Kleitman, Nathaniel (1962). Sleep and Wakefullness ed 2. Chicago: University of Chicago Press.
- ↑ Dijk DJ, Czeisler CA (January 1994). "Paradoxical timing of the circadian rhythm of sleep propensity serves to consolidate sleep and wakefulness in humans". Neuroscience Letters 166 (1): 63–8. doi:10.1016/0304-3940(94)90841-9. PMID 8190360.
- ↑ Dijk DJ, Czeisler CA (May 1995). "Contribution of the circadian pacemaker and the sleep homeostat to sleep propensity, sleep structure, electroencephalographic slow waves, and sleep spindle activity in humans". The Journal of Neuroscience 15 (5 Pt 1): 3526–38. PMID 7751928. http://www.jneurosci.org/cgi/pmidlookup?view=long&pmid=7751928.
- ↑ Cromie, William J. (1999-07-15). "Human Biological Clock Set Back an Hour". The Harvard University Gazette. http://www.hno.harvard.edu/gazette/1999/07.15/bioclock24.html. Retrieved 2008-02-19.
- ↑ Aldrich, Michael S (1999). Sleep medicine. New York: Oxford University Press. ISBN 0-19-512957-1. http://books.google.com.au/books?id=1jScwMrsmAMC&pg=RA1-PA65&lpg=RA1-PA65&dq=experimenting+with+the+28+hour+day&source=bl&ots=9R4mo2fI1O&sig=om2zbYPnXnm_1HuZo2Tch6J1vyo&hl=en&ei=MBZeStGgIoyJkQWd17znDA&sa=X&oi=book_result&ct=result&resnum=2.
- ↑ Pilcher JJ, Michalowski KR, Carrigan RD (2001). "The prevalence of daytime napping and its relationship to nighttime sleep". Behavioral Medicine 27 (2): 71–6. doi:10.1080/08964280109595773. PMID 11763827.
- ↑ Emily Rolston, Judy R. Sandlin, Michael Sandlin, and Rosanne Keathley (2007). "Power-Napping: Effects on Cognitive Ability and Stress Levels Among College Students". Power-Napping: Effects on Cognitive Ability and Stress Levels Among College Students. Liberty University. http://aahperd.confex.com/aahperd/2007/finalprogram/paper_10353.htm. Retrieved 2008-11-11.
- ↑ Sabah Quraishi (2007). "Circadian Rhythms and Sleep". Circadian Rhythms and Sleep. Serendip. http://serendip.brynmawr.edu/bb/neuro/neuro01/web1/Quirashi.html. Retrieved 2007-09-19.
- ↑ Sinert, Richard; Peter R Peacock, Jr (May 10, 2006). "Renal Failure, Acute". eMedicine from WebMD. http://www.emedicine.com/emerg/topic500.htm. Retrieved 2008-08-03.
- ↑ Figueiro MG, Bierman A, Plitnick B, Rea MS (2009). "Preliminary evidence that both blue and red light can induce alertness at night". BMC Neuroscience 10: 105. doi:10.1186/1471-2202-10-105. PMID 19712442.
- ↑ Figueiro MG, Rea MS, Bullough JD (2006). "Does architectural lighting contribute to breast cancer?". Journal of Carcinogenesis 5: 20. doi:10.1186/1477-3163-5-20. PMID 16901343.
- ↑ Sloane PD, Figueiro MG, Cohen L. 2008. Light Therapy for Sleep Disorders and Depression in Older Adults. Clinical Geriatrics, March 2008:2-8.
- ↑ http://www.aviationweek.com/aw/jsp_includes/articlePrint.jsp?storyID=news/FATIGex.xml&headLine=null Aviation Week Article
- ↑ http://aeromedical.org/Articles/Pilot_Fatigue.html Pilot Fatigue Study
- ↑ http://www.cnn.com/2009/TRAVEL/05/15/pilot.fatigue.buffalo.crash/index.html CNN Article
- ↑ Yin L, Wang J, Klein PS, Lazar MA (February 2006). "Nuclear receptor Rev-erbalpha is a critical lithium-sensitive component of the circadian clock". Science 311 (5763): 1002–5. doi:10.1126/science.1121613. PMID 16484495. Lay summary – National Institute of Mental Health (February 17, 2006).
- ↑ Martino TA, Oudit GY, Herzenberg AM, et al. (May 2008). "Circadian rhythm disorganization produces profound cardiovascular and renal disease in hamsters". American Journal of Physiology. Regulatory, Integrative and Comparative Physiology 294 (5): R1675–83. doi:10.1152/ajpregu.00829.2007. PMID 18272659.
- ↑ Straif K, Baan R, Grosse Y, et al. (December 2007). "Carcinogenicity of shift-work, painting, and fire-fighting". The Lancet Oncology 8 (12): 1065–6. doi:10.1016/S1470-2045(07)70373-X. PMID 19271347. Lay summary – WebMD (November 30, 2007).
- ↑ Uz T, Akhisaroglu M, Ahmed R, Manev H (December 2003). "The pineal gland is critical for circadian Period1 expression in the striatum and for circadian cocaine sensitization in mice". Neuropsychopharmacology 28 (12): 2117–23. doi:10.1038/sj.npp.1300254. PMID 12865893.
- ↑ Kurtuncu M, Arslan AD, Akhisaroglu M, Manev H, Uz T (April 2004). "Involvement of the pineal gland in diurnal cocaine reward in mice". European Journal of Pharmacology 489 (3): 203–5. doi:10.1016/j.ejphar.2004.03.010. PMID 15087244.
- ↑ McClung CA, Sidiropoulou K, Vitaterna M, et al. (June 2005). "Regulation of dopaminergic transmission and cocaine reward by the Clock gene". Proceedings of the National Academy of Sciences of the United States of America 102 (26): 9377–81. doi:10.1073/pnas.0503584102. PMID 15967985.
External links
- Circadian rhythm at the Open Directory Project
- Forger D, Gonze D, Virshup D, Welsh DK (June 2007). "Beyond intuitive modeling: combining biophysical models with innovative experiments to move the circadian clock field forward". Journal of Biological Rhythms 22 (3): 200–10. doi:10.1177/0748730407301823. PMID 17517910.
- Rodrigo G, Carrera J, Jaramillo A (2007). "Evolutionary mechanisms of circadian clocks". Central European Journal of Biology 2 (2): 233–253. doi:10.2478/s11535-007-0016-z.
- Lighting Research Center, Light & Health Program