Phosgene
| Phosgene | |
|---|---|
 |
|
 |
|
| IUPAC name | Dichloromethanal Carbonyl chloride |
| Other names | Phosgene; CG; carbon dichloride oxide; carbon oxychloride; carbonyl dichloride; chloroformyl chloride; dichloroformaldehyde; dichloromethanone |
| Identifiers | |
| CAS number | 75-44-5 |
| RTECS number | SY5600000 |
| SMILES |
|
| Properties | |
| Molecular formula | CCl2O |
| Molar mass | 98.9 g mol-1 |
| Appearance | colorless gas |
| Density | 4.248 g dm-3, gas (15 °C) |
| Melting point |
−118 °C (155 K) |
| Boiling point |
8 °C (281 K) |
| Solubility in water | hydrolysis |
| Structure | |
| Molecular shape | Planar, trigonal |
| Dipole moment | 1.17 D |
| Hazards | |
| MSDS | http://www.vngas.com/pdf/g67.pdf |
| NFPA 704 |
 0
4
1
|
| Flash point | non-flammable |
| Related compounds | |
| Related compounds | formaldehyde; carbonic acid; urea; carbon monoxide; chloroformic acid |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Infobox references |
|
Phosgene is the chemical compound with the formula COCl2. This colorless gas gained infamy as a chemical weapon during World War I, but it is also a valued industrial reagent and building block in organic synthesis. In low concentrations, its odor resembles freshly cut hay or grass. Some soldiers during the First World War have stated that it smelled faintly of May Blossom. In addition to its industrial production, small amounts occur naturally from the breakdown of chlorinated compounds and the combustion of chlorine-containing organic compounds.[1]
Contents |
Structure and basic properties
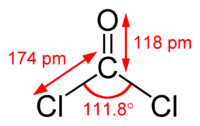
Phosgene is a planar molecule as predicted by VSEPR theory. The C=O distance is 1.18 Å, the C---Cl distance is 1.74 Å and the Cl---C---Cl angle is 111.8°.[2] It is one of the simplest acid chlorides, being formally derived from carbonic acid.
Production
Industrially, phosgene is produced by passing purified carbon monoxide and chlorine gas through a bed of porous activated carbon, which serves as a catalyst. The equation is described as follows:[1]
- CO + Cl2 → COCl2 ΔHrxn = −107.6kJ/mol
The reaction is exothermic, therefore the reactor must be cooled. Typically, the reaction is conducted between 50 and 150 °C because above 200 °C, phosgene reverts to carbon monoxide and chlorine, Keq (300K) = 0.05M. Approximately 5000 tonnes were produced in 1989.
Because of safety issues, phosgene is almost always produced and consumed within the same plant and extraordinary measures are made to contain this toxic gas. It is listed on schedule 3 of the Chemical Weapons Convention: all production sites manufacturing more than 30 tonnes per year must be declared to the OPCW.[3] Although much less dangerous than most other chemical weapons (e.g. mustard gas), phosgene is still regarded as a viable chemical warfare agent because it is so easy to manufacture when compared to the production requirements of more technically advanced chemical weapons such as the first-generation nerve agent tabun.
Adventitious occurrence
Upon ultraviolet radiation in the presence of oxygen, chloroform slowly converts into phosgene via a radical reaction. To suppress this photodegradation, chloroform is often stored in brown-tinted glass containers. Chlorinated compounds used to clean oils off of metals may also react under the UV created in a welding arc to produce phosgene.
Uses
The great majority of phosgene is used in the production of isocyanates, the most important being toluene diisocyanate (TDI) and methylene diphenyl diisocyanate (MDI). These isocyanates are precursors to polyurethanes. Significant amounts are also used in the production of polycarbonates via its reaction with bisphenol A.[1] Polycarbonates are an important class of engineering thermoplastic found, for example, in lenses in eye glasses.
Organic synthesis
Although phosgene still finds use in organic synthesis, a variety of substitutes have been developed, notably trichloromethyl chloroformate (“diphosgene”), which is a liquid at room temperature, and bis(trichloromethyl) carbonate (“triphosgene”), a crystalline substance.[4] Following are the three most useful reactions involving phosgene.
Synthesis of carbonates
Diols react with phosgene to give either linear or cyclic carbonates (R = H, alkyl, aryl):
- HOCR2-X-CR2OH + COCl2 → 1/n [OCR2-X-CR2OC(O)-]n + 2 HCl
Synthesis of isocyanates
The synthesis of isocyanates from amines illustrates the electrophilic character of this reagent and its use in introducing the equivalent of "CO2+" (R = alkyl, aryl): [5]
- RNH2 + COCl2 → RN=C=O + 2 HCl
Such reactions are conducted in the presence of a base such as pyridine that absorbs the hydrogen chloride.
Synthesis of acid chlorides
It is also used to produce acid chlorides from carboxylic acids:
- RCO2H + COCl2 → RC(O)Cl + HCl + CO2
Such acid chlorides react with amines and alcohols to give, respectively, amides and esters, which are commonly used intermediates. Thionyl chloride is more commonly and more safely employed for this application. A specific application for phosgene is the production of chloroformic esters:
- ROH + COCl2 → ROC(O)Cl + HCl
Inorganic chemistry
Although it is somewhat hydrophobic, phosgene reacts with water to release hydrogen chloride and carbon dioxide:
- COCl2 + H2O → CO2 + 2 HCl
Analogously, with ammonia, one obtains urea:
- COCl2 + 4 NH3 → CO(NH2)2 + 2 NH4Cl
Halide exchange with nitrogen trifluoride and aluminium tribromide gives COF2 and COBr2, respectively.[1]
History
Phosgene was synthesized by the chemist John Davy (1790-1868) in 1812 by exposing a mixture of carbon monoxide and chlorine to sunlight. He named it in reference to use of light to promote the reaction; from Greek, phos (light) and gene (born).[6] It gradually became important in the chemical industry as the 19th century progressed, particularly in dye manufacturing.
Chemical warfare
- Further information: Use of poison gas in World War I and Second Italo-Abyssinian War
Following the extensive use of phosgene gas in combat during World War I, it was stockpiled by various countries as part of their secret chemical weapons programs.[7][8][9]
Bhopal disaster
- Further information: Bhopal disaster
In 1984 in Bhopal, India 43 tonnes of methylisocyanate was accidentally released, killing or injuring tens of thousands of people. One of the components of the leak was probably phosgene.
Safety
Phosgene is an insidious poison as the odor may not be noticed and symptoms may be slow to appear.[10] Phosgene can be detected at 0.4 ppm, which is 4x the Threshold Limit Value. Its high toxicity arises, not from hydrogen chloride released by hydrolysis, but by the action of the phosgene on the proteins in the pulmonary alveoli. The alveoli are the site of gas exchange, and their damage disrupts the blood – air barrier and causing suffocation. Phosgene detection badges are worn by those at risk of exposure.[1]
Sodium bicarbonate may be used to neutralise liquid spills of phosgene. Gaseous spills may be mitigated with ammonia.[11]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 Wolfgang Schneider and Werner Diller "Phosgene" in Ullmann's Encyclopedia of Industrial Chemistry Wiley-VCH, Weinheim, 2002. DOI: 10.1002/14356007.a19_411. Article Online Posting Date: June 15, 2000
- ↑ Nakata, M.; Kohata, K.; Fukuyama, T.; Kuchitsu, K. (1980). "Molecular Structure of Phosgene as Studied by Gas Electron Diffraction and Microwave Spectroscopy. The rz Structure and Isotope Effect". Journal of Molecular Spectroscopy 83: 105–117. doi:.
- ↑ Annex on Implementation and Verification ("Verification Annex")
- ↑ Hamley, P. "Phosgene" Encyclopedia of Reagents for Organic Synthesis, 2001 John Wiley, New York. doi: 10.1002/047084289X.rp149
- ↑ R. L. Shriner, W. H. Horne, and R. F. B. Cox (1943). "p-Nitrophenyl Isocyanate". Org. Synth.; Coll. Vol. 2: 453.
- ↑ John Davy (1812). "On a Gaseous Compound of Carbonic Oxide and Chlorine". Philosophical Transactions of the Royal Society of London 102: 144–151. doi:. http://links.jstor.org/sici?sici=0261-0523%281812%29102%3C144%3AOAGCOC%3E2.0.CO%3B2-1.
- ↑ http://lithgow.yourguide.com.au/news/local/news/general/chemical-warfare-left-its-legacy/1237570.aspx
- ↑ http://lithgow.yourguide.com.au/news/local/news/general/chemical-warfare-left-its-legacy/1266856.aspx
- ↑ [1]
- ↑ Borak J., Diller W. F. (2001). "Phosgene exposure: mechanisms of injury and treatment strategies". Journal of Occupational and Environmental Medicine 43 (2): 110–9. doi:. PMID 11227628.
- ↑ "Phosgene: Health and Safety Guide". International Programme on Chemical Safety (1998).
External links
- Davy's account of his discovery of phosgene
- International Chemical Safety Card 0007
- NIOSH Pocket Guide to Chemical Hazards
- U.S. CDC Emergency Preparedness & Response
- U.S. EPA Acute Exposure Guideline Levels
- Regime For Schedule 3 Chemicals And Facilities Related To Such Chemicals, OPCW website
- CBWInfo website
- Use of Phosgene in WWII and in modern-day warfare (Refer to Section 4.C of the article)
- http://www.vngas.com/pdf/g67.pdf
|
||||||||||||||||||||