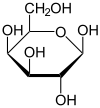
Galactose
| Galactose | |
|---|---|
 |
 |
| Identifiers | |
| CAS number | 26566-61-0 |
| PubChem | |
| MeSH | |
| Properties | |
| Molecular formula | C6H12O6 |
| Molar mass | 180.08 |
| Melting point |
167 degrees C |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Infobox references |
|
Galactose (Gal) (also called brain sugar[1]) is a type of sugar which is less sweet than glucose. It is considered a nutritive sweetener because it has food energy.
Galactan is a polymer of the sugar galactose. It is found in hemicellulose and can be converted to galactose by hydrolysis. Galactose solubility in water is 68.30 grams per 100 grams of water at 20–25° C.
Contents |
Sources
It is found in dairy products, in sugar beets and other gums and mucilages.
It is also synthesized by the body, where it forms part of glycolipids and glycoproteins in several tissues.
Relationship to lactose
Galactose is a monosaccharide constituent, together with glucose, of the disaccharide lactose. The hydrolysis of lactose to glucose and galactose is catalyzed by the enzyme lactase, a β-galactosidase. In the human body, glucose is changed into galactose in order to enable the mammary glands to secrete lactose.
Galactose and glucose are produced by hydrolysis of lactose by β-galactosidase. This enzyme is produced by the lac operon in Escherichia coli (E. coli).
Clinical significance
Two studies have suggested a possible link between galactose in milk and ovarian cancer.[2][3] Other studies show no correlation, even in the presence of defective galactose metabolism.[4][5] More recently, pooled analysis done by the Harvard School of Public Health showed no specific correlation between lactose containing foods and ovarian cancer, and showed statistically insignificant increases in risk for consumption of lactose at ≥30 g/d.[6] More research is necessary to ascertain possible risks.
There are some ongoing studies which suggest that galactose may have a role in treatment of focal segmental glomerulosclerosis (a kidney disease resulting in kidney failure and proteinuria). This effect is likely to be a result of binding of galactose to FSGS factor.
Structure and isomerism
The first and last -OH groups point the same way and the second and third -OH groups point the other way. D-Galactose has the same configuration at its penultimate carbon as D-glyceraldehyde. Galactose is a epimer of glucose.
Liver galactose metabolism
In the liver, galactose is converted to glucose 6-phosphate in the following reactions:
galacto- uridyl phosphogluco-
kinase transferase mutase
gal --------> gal 1 P ------------------> glc 1 P -----------> glc 6 P
^ \
/ v
UDP-glc UDP-gal
^ /
\___________/
epimerase
Metabolic disorders
There are 3 important disorders involving galactose:
| Name | Enzyme | Description |
| Galactosemia | Galactokinase | Causes cataracts and mental retardation. If a galactose-free diet starts sufficiently early, the cataracts will regress without complications however neurological damage is permanent. |
| UDPgalactose-4-epimerase deficiency | UDPgalactose-4-epimerase | Is extremely rare (only 2 reported cases). It causes nerve deafness. |
| Galactose-1-phosphate uridyl transferase deficiency | Galactose-1-phosphate uridyl transferase | Is the most problematic, as galactose-free diets do not have considerable long-term effects. |
References
- ↑ Compact American Medical Dictionary. Houghton Mifflin Reference Books (1998). ISBN 0395884098
- ↑ Cramer D (1989). "Lactase persistence and milk consumption as determinants of ovarian cancer risk". Am J Epidemiol 130 (5): 904–10. PMID 2510499.
- ↑ Cramer D, Harlow B, Willett W, Welch W, Bell D, Scully R, Ng W, Knapp R (1989). "Galactose consumption and metabolism in relation to the risk of ovarian cancer". Lancet 2 (8654): 66–71. doi:. PMID 2567871.
- ↑ Marc T. Goodman , Anna H. Wu , Ko-Hui Tung , Katharine McDuffie , Daniel W. Cramer , Lynne R. Wilkens , Keith Terada , Juergen K. V. Reichardt , and Won G. Ng (2002). "Association of Galactose-1-Phosphate Uridyltransferase Activity and N314D Genotype with the Risk of Ovarian Cancer". Am. J. Epidemiol 156 (8): 693–701. doi:. PMID 12370157.
- ↑ Fung, W. L. Alan, Risch, Harvey, McLaughlin, John, Rosen, Barry, Cole, David, Vesprini, Danny, Narod, Steven A. (2003). "The N314D Polymorphism of Galactose-1-Phosphate Uridyl Transferase Does Not Modify the Risk of Ovarian Cancer". Cancer Epidemiol Biomarkers Prev 12 (7): 678–80. PMID 12869412.
- ↑ Genkinger, Jeanine M., Hunter, David J., Spiegelman, Donna, Anderson, Kristin E., Arslan, Alan, Beeson, W. Lawrence, Buring, Julie E., Fraser, Gary E., Freudenheim, Jo L., Goldbohm, R. Alexandra, Hankinson, Susan E., Jacobs, David R., Jr., Koushik, Anita, Lacey, James V., Jr., Larsson, Susanna C., Leitzmann, Michael, McCullough, Marji L., Miller, Anthony B., Rodriguez, Carmen, Rohan, Thomas E., Schouten, Leo J., Shore, Roy, Smit, Ellen, Wolk, Alicja, Zhang, Shumin M., Smith-Warner, Stephanie A. (2006). "Dairy Products and Ovarian Cancer: A Pooled Analysis of 12 Cohort Studies". Cancer Epidemiol Biomarkers Prev 15: 364–372. doi:. PMID 16492930.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||