Atomic nucleus
The nucleus of an atom is the very dense region, consisting of nucleons (protons and neutrons), at the center of an atom. Although the size of the nucleus varies considerably according to the mass of the atom, the size of the entire atom is comparatively constant. Almost all of the mass in an atom is made up from the protons and neutrons in the nucleus with a very small contribution from the orbiting electrons.
The diameter of the nucleus is in the range of 1.6 fm (1.6 × 10−15 m) (for a proton in light hydrogen) to about 15 fm (for the heaviest atoms, such as uranium). These dimensions are much smaller than the size of the atom itself by a factor of about 23,000 (uranium) to about 145,000 (hydrogen).
The branch of physics concerned with studying and understanding the atomic nucleus, including its composition and the forces which bind it together, is called nuclear physics.
Contents |
Introduction
Etymology
The term nucleus is from Latin nucleus (“‘kernel’”), derived from nux (“‘nut’”). In 1844, Michael Faraday used the term to refer to the "central point of an atom". The modern atomic meaning was proposed by Ernest Rutherford in 1912.[1] The adoption of the term "nucleus" to atomic theory, however, was not immediate. In 1916, for example, Gilbert N. Lewis stated, in his famous article The Atom and the Molecule, that "the atom is composed of the kernel and an outer atom or shell".

Nuclear makeup
The nucleus of an atom consists of protons and neutrons (two types of baryons) bound by the nuclear force (also known as the residual strong force). These baryons are further composed of subatomic fundamental particles known as quarks bound by the strong interaction. Which chemical element an atom represents is determined by the number of protons in the nucleus. Each proton carries a single positive charge, and the total electrical charge of the nucleus is spread fairly uniformly throughout its body, with the exception of hydrogen and helium, where the charge is concentrated most highly at the central point, as would be expected for fermions in 1s states without orbital angular momentum.[2] As each proton carries a unit of charge, the charge distribution is indicative of the proton distribution, and the neutron distribution probably is similar.[2]
Protons and neutrons
Protons and neutrons are fermions, with different values of the isospin quantum number, so two protons and two neutrons can share the same space wave function. They sometimes are viewed as two different states of the same particle, the nucleon.[3][4]
In the rare case of a hypernucleus, a third baryon called a hyperon, with a different value of the strangeness quantum number can also share the wave function. However, the latter type of nuclei are extremely unstable and are not found on Earth except in high energy physics experiments.
The neutron has a positively charged core of radius ≈ 0.3 fm surrounded by a compensating negative charge of radius between 0.3 fm and 2 fm. The proton has an approximately exponentially decaying charge distribution with a mean square radius of about 0.8 fm.[5]
Forces
Nuclei are bound together by the residual strong force. The residual strong force is minor residuum of the strong force which binds quarks together to form protons and neutrons. This force is much weaker between neutrons and protons because it is mostly neutalized within them, in the same way that electromagnetic forces between neutral atoms (van der Waals forces) are much weaker than the electromagnetic forces that hold the atoms internally together.
The strong force is so named because it is significantly larger in magnitude than the other fundamental forces (electroweak, electromagnetic and gravitational). The strong force is highly attractive at very small distances, and this overwhelms the repulsion between protons due to the electromagnetic force, thus allowing nuclei to exist. However, because the residual strong force has a limited range because it decays quickly with distance (see Yukawa potential), only nuclei smaller than a certain size can be completely stable. The largest known complete stable nucleus is lead-208 which contains 208 neutrons and protons. Nuclei larger than this maximal size of 208 particles generally become increasingly short-lived, as the number of neutrons and protons which compose them increases beyond this number.
The residual strong force usually acts over a very short range (a few fermis, roughly one or two nucleon diameters) and causes an attraction between nucleons (protons and neutrons). However there are also halo nuclei such as lithium-11 or boron-14, in which dineutrons, or other collections of nucleons, orbit at distances of about ten fermis (similar to the size of lead-208). Such nuclei are always short-lived; for example, lithium-11 has a half-life of less than 8.6 milliseconds.
Nuclear models
The nuclear radius (R) is considered to be one of the basic things that any model must explain. It is roughly proportional to the cube root of the mass number (A) of the nucleus, and particularly in nuclei containing many nucleons, as they arrange in more spherical configurations:
The nucleus has approximately a constant density and therefore the nuclear radius R can be approximated by the following formula,
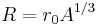
where, A = Atomic mass number (the number of protons, Z, plus the number of neutrons, N) and r0 = 1.25 fm = 1.25 × 10−15 m. In this equation, the constant r0 varies by 0.2 fm, depending on the nucleus in question, but this is less than 20% change from a constant. [6]
In other words, packing protons and neutrons in the nucleus gives approximately the same total size result as packing hard spheres of a constant size (like marbles) into a tight bag.
Liquid drop models
Early models of the nucleus viewed the nucleus as a rotating liquid drop. In this model, the trade-off of long-range electromagnetic forces and relatively short-range nuclear forces, together cause behavior which resembled surface tension forces in liquid drops of different sizes. This formula is successful at explaining many important phenomena of nuclei, such as their changing amounts of binding energy as their size and composition changes (see semi-empirical mass formula), but it does not explain the special stability which occurs when nuclei have special "magic numbers" of protons or neutrons.
Shell models and other quantum models
A number of models for the nucleus have also been proposed in which nucleons occupy orbitals, much like the atomic orbitals in atomic physics theory. These wave models imagine nucleons to be either sizeless point particles in potential wells, or else probability waves as in the "optical model", frictionlessly orbiting at high speed in potential wells.
In these models, the nucleons occupy orbitals in pairs, due to being fermions, but the exact nature and capacity of nuclear shells differs somewhat from those of elections in atomic orbitals, primarily because the potential well in which the nucleons move (especially in larger nuclei) is quite different from the central electromagnetic potential well which binds electrons in atoms. Nevertheless, the resemblance to atomic orbital models may be seen in a small atomic nucleus like that of helium-4, in which the two protons and two neutrons separately occupy 1s orbitals analogous to the 1s orbitals for the two electrons in the helium atom, and achieve unusual stability for the same reason. This stability also underlies the fact that nuclei with 5 nucleons are all extremely unstable and short-lived.
For larger nuclei, the shells occupied by nucleons begin to differ significantly from electron shells, but nevertheless, present nuclear theory does predict the "magic numbers" of filled nuclear shells for both protons and neutrons. The closure of the stable shells predicts unusually stable configurations, analogous to the noble group of nearly-inert gases in chemistry. An example is the stability of the closed shell of 50 protons, which allows tin to have 10 stable isotopes, more than any other element. Similarly, the distance from shell-closure explains the unusual instability of isotopes which have far from stable numbers of these particles, such as the radioactive elements 43 (technetium) and 61 (promethium), each of which is preceded and followed by 17 or more stable elements.
Consistency between models
As with the case of superfluid liquid helium, atomic nuclei are an example of a state in which both (1) "ordinary" particle physical rules for volume and (2) non-intuitive quantum mechanical rules for a wave-like nature apply. In superfluid helium, the helium atoms have volume, and essentially "touch" each other, yet at the same time exhibit strange bulk properties, consistent with a Bose-Einstein condensation. The latter reveals that they also have a wave-like nature and do not exhibit standard fluid properties, such as friction. For nuclei made of hadrons which are fermions, the same type of condensation does not occur, yet nevertheless, many nuclear properties can only be explained similarly by a combination of properties of particles with volume, in addition to the frictionless motion characteristic of the wave-like behavior of objects trapped in Schroedinger quantum orbitals.
See also
- Atomic mass
- Atomic number
- Giant resonance
- Isotope
- Liquid drop model
- List of particles
- Nuclear fusion
- Nuclear fission
- Nuclear medicine
- Nuclear physics
- Nuclear structure
- Radioactivity
- Semi-empirical mass formula
References
- ↑ Nucleus – Online Etymology Dictionary
- ↑ 2.0 2.1 Jean-Louis Basdevant, James Rich & Michel Spiro (2005). Fundamentals in Nuclear Physics. Springer. p. Fig. 1.1, p. 13. ISBN 0387016724. http://books.google.com/books?id=OFx7P9mgC9oC&pg=PA375&dq=helium+%22nuclear+structure%22&lr=&as_brr=0#PPA13,M1.
- ↑ A. G. Sitenko, Viktor Konstantinovich Tartakovskiĭ (1997). Theory of Nucleus: Nuclear Structure and Nuclear Interaction. p. p. 3. ISBN 0792344235. http://books.google.com/books?id=swb9QpqOqtAC&pg=PA464&dq=isbn:0792344235#PPA3,M1.
- ↑ Mark Allen Srednicki (2007). Quantum Field Theory. Cambridge University Press. p. pp. 522-523. ISBN 9780521864497. http://www.worldcat.org/search?q=9780521864497&qt=owc_search.
- ↑ Jean-Louis Basdevant, James Rich & Michel Spiro. Fundamentals in Nuclear Physics. p. p. 155. http://books.google.com/books?id=OFx7P9mgC9oC&pg=PA375&dq=helium+%22nuclear+structure%22&lr=&as_brr=0#PPA155,M1.
- ↑ Literature: Krane, Kenneth S., Introductory Nuclear Physics, Wiley, 1987.
External links
|
||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||