Aripiprazole
 |
|
|
Aripiprazole
|
|
| Systematic (IUPAC) name | |
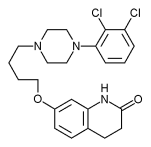
| 7-[4-[4-(2,3-dichlorophenyl) piperazin-1-yl]butoxy]- 3,4-dihydro-1H-quinolin-2-one |
|
| Identifiers | |
| CAS number | |
| ATC code | N05 |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | C23H27Cl2N3O2 |
| Mol. mass | 448.385 |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | 87% |
| Protein binding | >99% |
| Metabolism | liver |
| Half life | 75h (active metabolite : 94h) |
| Excretion | feces and urine |
| Therapeutic considerations | |
| Licence data |
, |
| Pregnancy cat. |
C (USA) |
| Legal status |
℞ Prescription only |
| Routes | oral (via tablets, orodispersable tablets, and oral solution); intramuscular |
Aripiprazole (ay-ree-PIP-ray-zole) (sold as Abilify) was approved by the Food and Drug Administration (FDA) on November 15, 2002 for the treatment of schizophrenia, the sixth atypical antipsychotic medication of its kind. More recently it received FDA approval for the treatment of acute manic and mixed episodes associated with bipolar disorder, and as an adjunct for the treatment of depression.[1] Aripiprazole was developed by Otsuka in Japan; in the U.S., Otsuka America markets the drug jointly with Bristol-Myers Squibb. Annual sales approx $2.1bn.
Contents |
Pharmacology
Aripiprazole's mechanism of action is different from the other FDA-approved atypical antipsychotics (e.g., clozapine, olanzapine, quetiapine, ziprasidone, and risperidone). Rather than antagonizing the D2 receptor, aripiprazole appears to be a D2 partial agonist[2] and selective agonist.[3] D2 and D3 receptor occupancy levels are high, with average levels ranging between ~71% at 2mg/day to ~96% at 40mg/day.[4] Most atypical antypsychotics bind preferentially to extrastriatal receptors, but aripiprazole appears to be less preferential in this regard, as binding rates are high throughout the brain.[5]
In addition to its functional selectivity at the D2 receptor, aripiprazole is also a partial agonist at the 5-HT1A receptor, and like the other atypical antipsychotics displays an antagonist profile at the 5-HT2A receptor. Aripiprazole has moderate affinity for histamine and alpha-adrenergic receptors and for the serotonin transporter, and no appreciable affinity for cholinergic muscarinic receptors.[6]
Its mechanism of action makes aripiprazole a useful adjunct in the short-term treatment of psychosis associated with MDMA abuse. It is also useful in the treatment of psychosis generated by compounds similar to MDMA and its derivatives. It is not advised in the treatment of the chemically similar, but non-derivative, paramethoxyamphetamine (PMA).[7]
Pharmacokinetics
Aripiprazole displays linear kinetics and has an elimination half-life of approximately 75 hours. Steady-state plasma concentrations are achieved in about 14 days. Cmax (maximum plasma concentration) is achieved 3-5 hours after oral dosing. Bioavailability of the oral tablets is about 90% and the drug undergoes extensive hepatic metabolization (dehydrogenation, hydroxylation, and N-dealkylation), principally by the enzymes CYP2D6 and CYP3A4. Its only known active metabolite is dehydro-aripiprazole, which typically accumulates to approximately 40% of the aripiprazole concentration.[6] Its elimination half-life is about 94 hours. The parenteral drug is excreted only in traces, and its metabolites, active or not, are excreted via feces and urine.[8]
Patent status and availability
Otsuka's patent on aripiprazole expires on October 20, 2014;[9] however, due to a pediatric extension, a generic will not become available until at least April 20, 2015.[10]
Aripiprazole is available in 2mg, 5mg, 10mg, 15mg, 20mg, and 30mg tablets.[11]Aripiprazole is also available as 10mg and 15mg orally-disintegrating tablets, 1mg/1ml solution and 7.5mg/ml intramuscular injection.
Warnings about medications with similar names
Warning flags are raised by the drug's name: the '-prazole' suffix incorrectly associates the drug with the proton pump inhibitors (such as omeprazole, pantoprazole, and lansoprazole), that are used to treat peptic ulcer disease. However, aripiprazole is in an entirely different class of drugs, and can provoke unnecessary side effects when incorrectly prescribed for peptic ulcer disease. Also, aripiprazole can be confused with "azole" antifungals, such as ketoconazole and clotrimazole.
Side effects
Common side effects: Akathisia, headache, unusual tiredness or weakness, nausea, vomiting, an uncomfortable feeling in the stomach, constipation, light-headedness, trouble sleeping, restlessness, sleepiness, shaking, and blurred vision.
Uncommon side effects: Uncontrollable twitching or jerking movements, tremors, seizure, and weight gain. Some people may feel dizzy, especially when getting up from a lying or sitting position, or may experience a fast heart rate.
Rare side effects: Combination of fever, muscle stiffness, faster breathing, sweating, reduced consciousness, and sudden change in blood pressure and heart rate (neuroleptic malignant syndrome).
Very rare side effects: Allergic reaction (such as swelling in the mouth or throat, itching, rash), increased production of saliva, speech disorder, nervousness, agitation, fainting, reports of abnormal liver test values, inflammation of the pancreas, muscle pain, weakness, stiffness, or cramps.
As with all antipsychotic medication, patients using aripiprazole may develop the permanent neurological disorder tardive dyskinesia.
While taking aripiprazole some elderly patients with dementia have suffered from stroke or 'mini' stroke. Other patients may experience high blood sugar or the onset or worsening of diabetes.
Drug interactions
Aripiprazole is a substrate of CYP2D6 and CYP3A4. Coadministration with medications that inhibit (e.g. paroxetine, fluoxetine) or induce (e.g. carbamazepine) these metabolic enzymes is known to increase and decrease, respectively, plasma levels of aripiprazole.[12]
Aripiprazole may change the subjective effects of alcohol. One study[13] found that aripiprazole increased the sedative effect and reduced the sense of euphoria normally associated with alcohol consumption. However, another study[14] found that there was no difference in subjective effect between a placebo group and a group taking aripiprazole.
See also
- Tardive dysphrenia
References
- ↑ FDA OKs Abilify for Depression
- ↑ Lawler CP et al. (1999). "Interactions of the novel antipsychotic aripiprazole (OPC-14597) with dopamine and serotonin receptor subtypes". Neuropsychopharmacology 20 (6): 612–27. doi:. PMID 10327430.
- ↑ Urban JD et al. (2007). "Aripiprazole has functionally selective actions at dopamine D2 receptor-mediated signaling pathways". Neuropsychopharmacology 32 (1): 67–77, 2007. doi:. PMID 16554739.
- ↑ Kegeles, LS et al. (2008). "Dose–Occupancy Study of Striatal and Extrastriatal Dopamine D2 Receptors by Aripiprazole in Schizophrenia with PET and [18F]Fallypride". Neuropsychopharmacology 33 (13): 3111-3125. doi:. PMID 18418366.
- ↑ "In This Issue". Am J Psychiatry 165 (8): A46. 2008. doi:.
- ↑ 6.0 6.1 Drug label - Clinical Pharmacology
- ↑ AJ Giannini. Drugs of Abuse--Second Edition. Los Angeles, Practice Management Information Corp., 1997.
- ↑ (WO/2007/035348) NANOPARTICULATE ARIPIPRAZOLE FORMULATIONS
- ↑ Oshiro, Yasuo; Seiji Sato & Nobuyuki Kurahashi, "Carbostyril derivatives", US 5006528, published October 20, 1989, issued April 9, 1991
- ↑ Electronic Orange Book – Patent and Exclusivity Info
- ↑ U.S. Food and Drug Administration Approves ABILIFY(R) (aripiprazole) for the Acute Treatment of Manic and Mixed Episodes Associated With Bipolar I Disorder in Pediatric Patients (10 to 17 Years of Age) - Forbes.com
- ↑ Drug label - Warnings and Precautions
- ↑ Kranzler, Henry R. et al (2008). "Effects of Aripiprazole on Subjective and Physiological Responses to Alcohol". Alcoholism: Clinical and Experimental Research 32 (4): 573-579. doi:. PMID 18261195.
- ↑ Konstantin Voronin, Patrick Randall, Hugh Myrick, Raymond Anton (2008). "Aripiprazole Effects on Alcohol Consumption and Subjective Reports in a Clinical Laboratory Paradigm—Possible Influence of Self-Control". Alcoholism: Clinical and Experimental Research 32 (11): 1954—1961. doi:. PMID 18782344.
External links
|
|||||||||||||||||||||||||||||||||||||||||